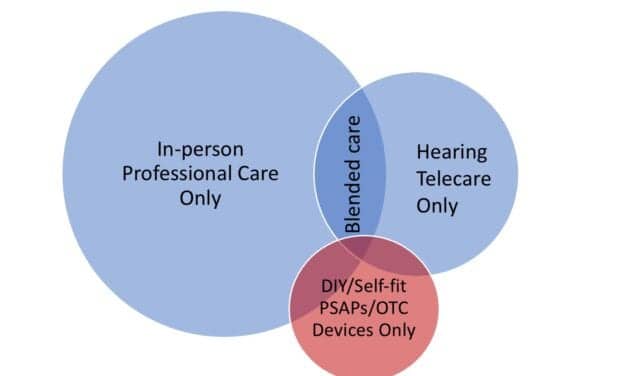
Nuheara Receives FDA Clearance for HP Hearing PRO
Following the OTC hearing aid law going into effect on October 17, 2022 in the US, Nuheara plans to bring to market its first medical device as the HP Hearing PRO under its worldwide Trademark License Agreement for use of HP trademarks on Nuheara’s hearing aids, personal sound amplification devices, and accessories.