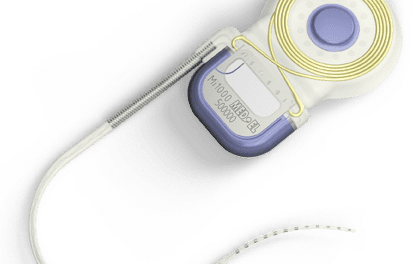
Vivosonic Inc creates products to make life a little easier for diagnostic clinicians.
Before Vivosonic grew to become a developer and manufacturer of novel audiological medical devices, the Toronto-based company was simply a research project.
Vivosonic President and CEO Yuri Sokolov, PhD, MBA, credits that project and, more specifically, the efforts of a man named Poul Madsen for the eventual creation of his company.
In 1996, Sokolov received the first Dr G.R. Heffernan/Co-Steel Innovation Scholarship from the University of Toronto’s Institute of Biomaterials and Biomedical Engineering (IBBME) for a research project titled “Design, testing, and commercialization study of an auditory mini-screener.” The purpose of the scholarship was to combine innovation with management, and technological research with commercialization.
At the time, Madsen—who was one of the most prominent industrialists and developers in diagnostic audiology equipment, having created the first commercial impedance audiometer, and the first commercial evoked response audiometer—was an adjunct professor at the university’s IBBME.
During the 3-year course of the research project, Sokolov and his collaborators developed a method of continuous real-time estimation and recording of physiological signals, Vivography™, Otoacoustic Emission Probe, and a prototype of a Distortion Product Otoacoustic Emissions (DPOAE) portable screener. That technology was to be transferred to Madsen’s company, Poul Madsen Medical Devices Ltd, for commercialization, but he died in 1997 before that could be accomplished.
“He was a great mind and a pioneer in diagnostic audiology equipment,” Sokolov says of Madsen. “He was a great part of the biomedical acoustics research group [at the university] and instrumental at the very early stage of this technology development. We are so thankful to Professor Madsen for his contribution and follow his pioneering path in creating the world’s first patented technologies and products.”
Creating Vivosonic
In 1999, Sokolov created Vivosonic to achieve the commercialization that Madsen no longer could, and to continue developing and manufacturing products to make the lives of diagnostic clinicians easier.
“We are focused on the technology that enables the clinicians to do diagnostic tests easier and faster, to eliminate frustrations that clinicians experience in the real world, and to make their work more efficient,” Sokolov says. “We do that through innovative technologies.

“Simply said, what we’re trying to do is to make the hard job that clinicians perform many hours every day a little bit more fun, a bit easier, and a bit better for them,” Sokolov says. “They’re so overloaded with their [number of] patients that they have little time to actually talk to [them] because much of their time is taken by those tests. Even if you look at the user interfaces of conventional devices, they’re so complex, and there’s so many things to learn, and there’s such an abundance of information that you have to really pick exactly what information is relevant, and which is exactly needed for a diagnosis.
“So we’re trying to make it easier for them, so they can spend more time with their patients, talking to them, analyzing results, and solving the patients’ problems, but not struggle with the equipment problems,” he says.
Since its formation, Vivosonic has focused on creating technologies to solve those problems. In 2000, the company developed its first product, the VivoScan™ with DPOAE function, which later received approval from the United States Food and Drug Administration (FDA) and Health Canada for sale, beginning in December 2001.
This past January, Vivosonic received FDA approval for its latest product, Integrity™, which it touts as the world’s first and only wireless objective hearing-assessment system. The Integrity features three other technologies created by Vivosonic: Amplitrode™, an electrode amplifier that reduces frustrating noises by amplifying electrical potentials in situ, directly on the ground electrode; VivoLink™, a wireless Bluetooth® interface between the signal-recording unit and the computer; and Vivography™, an advanced digital signal-processing technique that reduces the patient’s muscular-motion artifacts and speeds up testing. The system expands the company’s objective hearing-assessment product range from otoacoustic emissions (OAEs) into auditory evoked potentials (AEPs): auditory brainstem response (ABR) and auditory steady state response (ASSR).
The Integrity is the result of a survey Vivosonic conducted in 2004 that asked 60 audiologists across the United States to identify and quantify clinicians’ problems with the performance of available electrophysiological postscreening hearing assessment in infants. The survey focused on ABR and ASSR.
“Among other things, we asked them to identify what frustrations, if any, they encountered when they do OAE and ABR and ASSR testing, and to rank those frustrations,” says Vivosonic’s product marketing manager Sidney Tannenbaum, MBA. “Our data came up with some interesting findings—that electromagnetic and physiological noise were among the highest frustrations that audiologists were experiencing when they were doing evoked potential testing. We also asked how they would rate a wireless feature in ABR and ASSR testing, and they gave very positive responses.
“We took those frustrations and desires from audiologists and translated that into an instrument, which we hope will increase the efficiency of audiologists and take away those frustrations,” Tannenbaum says.
Sokolov says the Integrity’s wireless capabilities did just that.
“On the one hand, it solved a problem of electrical noise coming from the power line. On the other hand, it provides the comfort of mobility to both the clinician and the patient. That not only reduces the frustration of the clinician, but also makes it much more comfortable for the caregiver when the child is being tested,” Sokolov says. “The parents who come back for a follow-up diagnosis after their child was screened positively for a suspected hearing loss come to this test already anxious. In the operating room, it gives much more freedom to the audiologist and the surgeon and the operating crew.”
The wireless features make the test less intimidating for patients—especially useful when working with pediatric patients.
“Many systems don’t even look friendly. They raise the anxiety of patients and the parents of young children who are being tested. So we also try to make the instruments look friendly,” Sokolov says. “When we were developing the Amplitrode, one of the major requirements was to have it look friendly to a parent who sees their child being tested.”
The Amplitrode has release buttons so that clinicians can more easily place the electrodes on patients and take them off, and make it more comfortable overall.
“Audiology is like any other medical field or even consumer products field; people want the tools that they use in their work to be much more user-friendly and more convenient than they were years ago,” Sokolov says. “Wireless communications are penetrating all areas of life, and I think audiology is no exception to that.
“Audiology is moving toward much more precise diagnostics. Years ago, there was sensorineural hearing loss, which was not distinguishing between the cochlea and the VIIIth nerve. Today, they need much more precise diagnostics because they have tools to treat those conditions—cochlear implants, hearing aids, implanted bone conduction devices, and other tools,” he says. “For these conditions, clinicians need objective electrophysiological diagnostic tools, in addition to, and sometimes in place of, subjective tests. That’s where we see our role, in these new testing technologies. Our role is not in developing the clinical or physiological part of it, but in the technological solution. That’s where we come in.”
Sokolov says Vivosonic’s “holistic approach” to creating new technologies sets the company apart from others in the industry.
“We looked at the problems that clinicians experience, and we analyzed and dissected them. We set a goal for ourselves to solve those problems, and that’s what we did,” Sokolov says. “It’s not only in situ amplification and wireless technology that we developed, but also very innovative signal-processing techniques.
“For the whole system to function really well, and to eliminate those frustrations, and enable the clinician to have flexibility and mobility, we took this approach of solving various problems, not just one,” he says. “Of course, first we had to analyze those problems, and that approach is quite different from what manufacturers typically do. They would normally acquire some research from a university or a clinical research group and sort of wrap it up in a new user interface or a recognizable brand-name enclosure of the manufacturer. It gives a nice look to the product, but the core technology remains the same, and of course it inherits all or most of the features of the research system that was a product of that university or research group, which is not necessarily good for regular clinical use for many reasons.

“What we did is say, ‘Let’s forget about everything that was technologically done in this respect and let’s create something that addresses the issues directly.’”
Vivosonic identifies potential new projects by looking for problems to solve. The company’s client base is made up primarily of audiologists and ENTs across the world.
“If there’s no problem a clinician experiences, we don’t think coming up with yet another device would be a success,” Sokolov says, “because if there’s no problem to fix, there’s not much of a demand.
“With this wireless solution that we came up with, it seems from our contacts at a series of recent conferences we’ve been to that there are other clinical applications for it that we didn’t even think about,” Sokolov says. “We started with the application of the Amplitrode and wireless communications to infant postscreening assessment, because ASSR has been used there. ASSR signal is so small, so faint that the test is pretty difficult to perform and lengthy. The results are not necessarily always precise enough. We looked into that problem first, and then when we developed this wireless solution and in situ amplification, we realized that it’s an enabling technology that can be applied to other signals. We are speaking with other medical professions that do other types of surgeries and diagnostic procedures where signals are also very small, very faint, and are contaminated with electromagnetic noises and difficult to record. This technology can be applied to other fields as well, and that’s probably something that we’ll be looking at.”
Among the potential uses for the Integrity are operating room applications, animal research applications, and ABR testing in a hyperbaric chamber. Other ABR instruments cannot be placed inside a hyperbaric chamber, but the wireless system offers a solution.
Tannenbaum says Vivosonic’s problem-solving method is the best way to ensure that the products it creates will be of use to health care professionals.
“The ideal way is not to take technology and say, ‘What can I do with it?’“ Tannenbaum says. “The ideal way is to see what the problems are in the market, what the needs are, and then say, ‘What technology is available or what can we develop to meet those needs?’
“You often have the opposite effect, where people might stumble on a technology or some innovation, and then go looking for a problem to solve. What we’re trying to do here is look at the industry that we’re in and see what problems there are, and then solve them in an innovative way,” Tannenbaum says.

Vivosonic is also involved in giving clinical courses, workshops, and seminars throughout the United States and other parts of the world.
“We see that as a way of educating the audiology community about the benefits of wireless communication and in situ amplification, and informing them of the benefits of Integrity,” Tannenbaum says.
Sokolov credits the “enthusiasm and spirit of innovation” among the company’s 15 employees as the driving force behind its products. Vivosonic has received two patents for its technologies, and has several patent applications pending.
“We have an excellent research and engineering team,” Sokolov says. “We’ve got an excellent production/operations team that builds systems to the highest quality standards.”
Vivosonic’s quality system is certified to ISO 13485, a quality management standard for medical devices.
Sokolov says Vivosonic’s future will be spent “looking for other problems.”
“You have to really ask the user in the market what they actually want, how they use it in the field, and make it part of the requirements, and that’s exactly what we did with the Integrity,” he says. “We keep learning. We keep talking to the clinicians. And we’ll definitely be adding more features as asked for and required by clinicians.”
And whenever problems arise?
“We’ll be there to solve them,” Sokolov says.
Danielle Cohen is associate editor of Hearing Products Report.