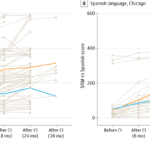
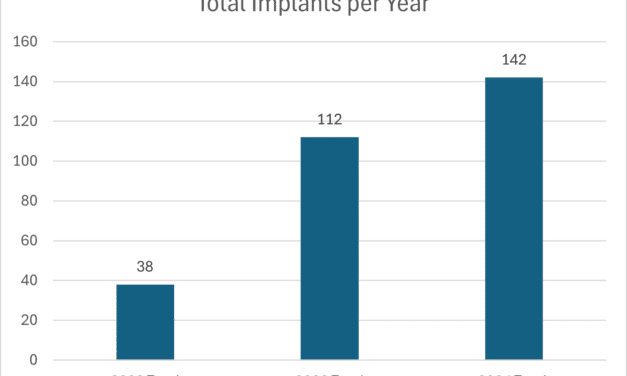
Providing Comprehensive Care
Hearing care professionals can provide more comprehensive patient support and expand practice revenue by offering guidance on hearing technologies, ancillary products, and emerging treatment options beyond traditional hearing aids.