With several completely implantable CIs in development, a new device choice for patients with severe-to-profound hearing loss is on the horizon.
By Melanie Hamilton-Basich
For decades, creating a feasible fully invisible cochlear implant (CI) has been a goal in the hearing care field. While traditional CIs have provided life-changing benefits, the necessity of an external sound processor has remained a barrier for some patients due to lifestyle limitations, maintenance, or social stigma. Now, with several manufacturers in or entering pivotal clinical trials, the era of the completely implantable cochlear implant is moving from concept to reality, promising new options for patients and new considerations for hearing care professionals.
These investigational devices aim to integrate all components—including the microphone, processor, and power source—under the skin, offering users the freedom to hear 24/7 without wearing any external equipment. Three companies in the implantable hearing space—Cochlear, Med-El, and Envoy Medical—are pursuing this technology, each with a unique design philosophy and approach. While all are still for investigational use only, the decades-long wait for this type of device is now nearing an end.
The potential impact extends beyond just invisibility. According to Caroline Arnedt, AuD, director of audiology at Envoy Medical, the prospect of a completely implantable CI is already attracting a new demographic of patients. “What’s interesting is what I’m hearing from the [trial] sites is that people who would have never set foot in a cochlear implant clinic previously, because they knew that the end result was going to have to be wearing this [external] device, are now coming in,” she says. “They’ll say, ‘I’m here because I want something that’s going to work with my lifestyle and it’s going to be just part of me.’”
Redefining the Patient Experience
For hearing care professionals, the conversation around CIs often involves managing patient expectations about the daily logistics of an external device.
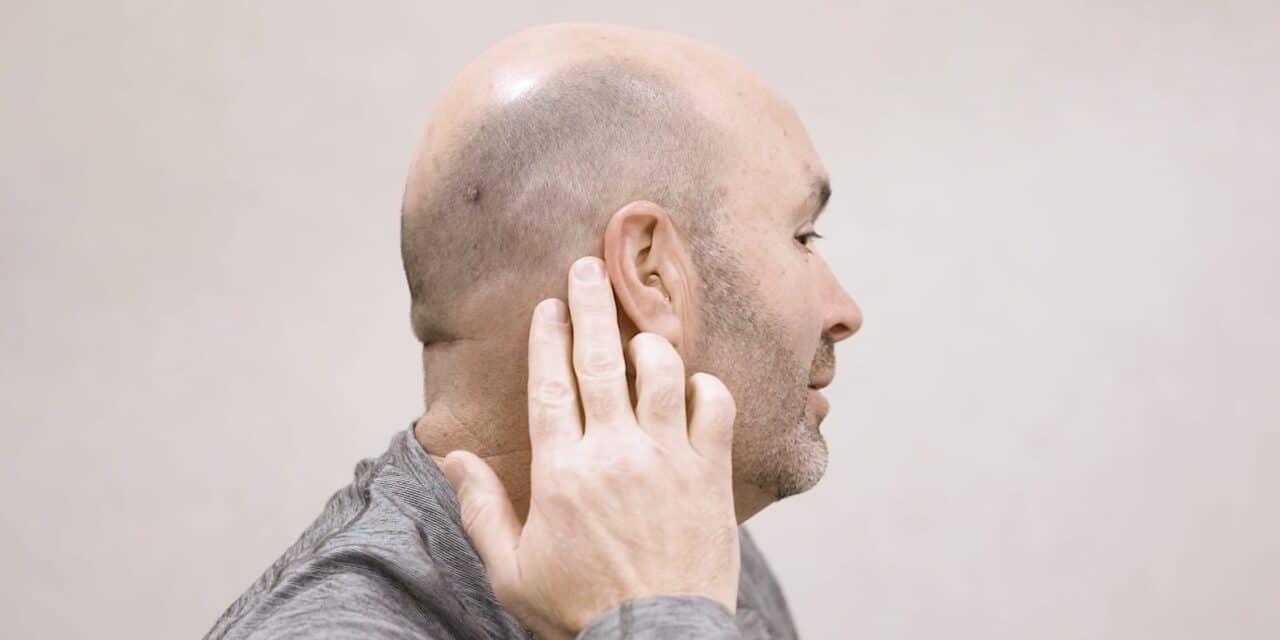
The primary appeal of a completely implantable cochlear implant is the ability to eliminate daily management of an external processor and the social stigma some patients feel because of it. This can be particularly meaningful for individuals with active lifestyles. For example, one clinical trial participant was able to go snorkeling in a tropical destination with Envoy Medical’s Acclaim device. “She was so excited to hear the water, to hear the waterfall, but even more so, she was there with a family member, and she was able to talk and interact with that family member while they were swimming,” Arnedt says. While waterproof covers exist for traditional CIs, she notes that patients often report they are not as acoustically transparent, and there is always a worry about the external processor falling off.
Another key benefit is the potential for 24/7 hearing. Since a completely implantable CI does not need to be removed at night, users can maintain auditory awareness while sleeping. This can provide a greater sense of safety and connection.
A Look at the Technology
While the goal of a fully invisible device is shared, the three companies are taking different paths to get there, resulting in distinct technological approaches.
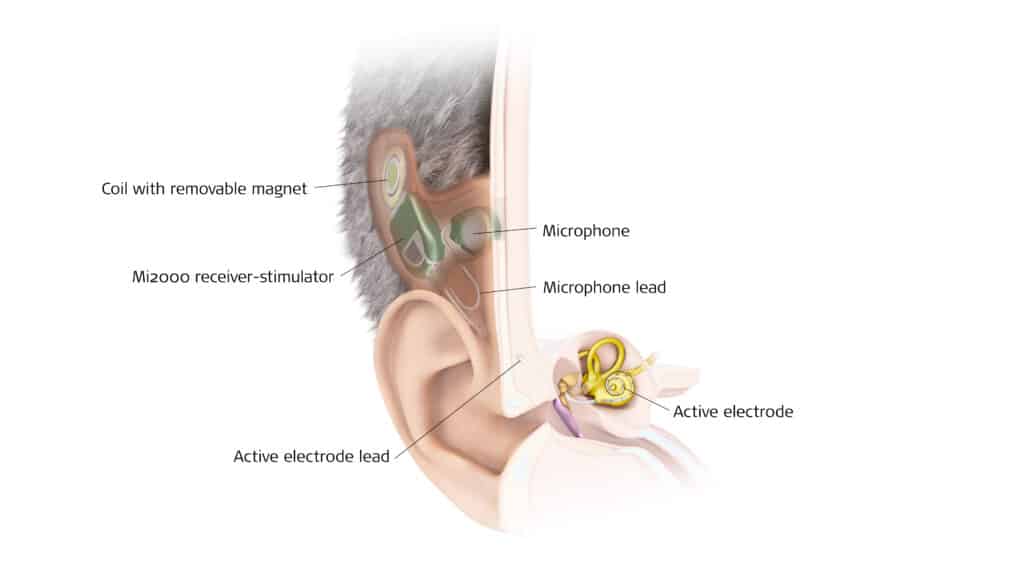
Cochlear is leveraging its 45 of experience, including 21 years researching fully implantable technology, which has led to its third-generation device. According to Jan Janssen, chief technology officer at Cochlear, their system is designed to integrate all key components, including the microphone and battery, under the skin behind the ear. This design is built upon the company’s existing implant designs and surgical procedures. A key focus of Cochlear’s long-term research has been the challenge of internal noise. “Cochlear’s two decades of fully implantable research have consistently shown that effective management of body-generated noise is critical to a positive user experience, and has informed our design principles over time,” Janssen says.
Med-El is also building upon its existing platform. “Our totally implantable cochlear implant system, or TICI, is built on more than 40 years of Med-El experience. It uses the same proven concepts you know from our traditional CI platform,” says Jennifer Robinson, corporate director of product management for Med-El.
Their system utilizes a new subcutaneous microphone and integrates their established signal processing approach including fine structure strategies that convey both envelope and fine structure cues. The TICI also utilizes Med-El’s unique broad portfolio of electrodes in different lengths to closely match the size of the cochlea for individualized placement and help retain as much hearing as possible across all frequencies. “With our TICI system, a recipient and a professional can expect great results like they’re used to seeing with our traditional CI, and this has been shown in the published feasibility study results,” Robinson adds.
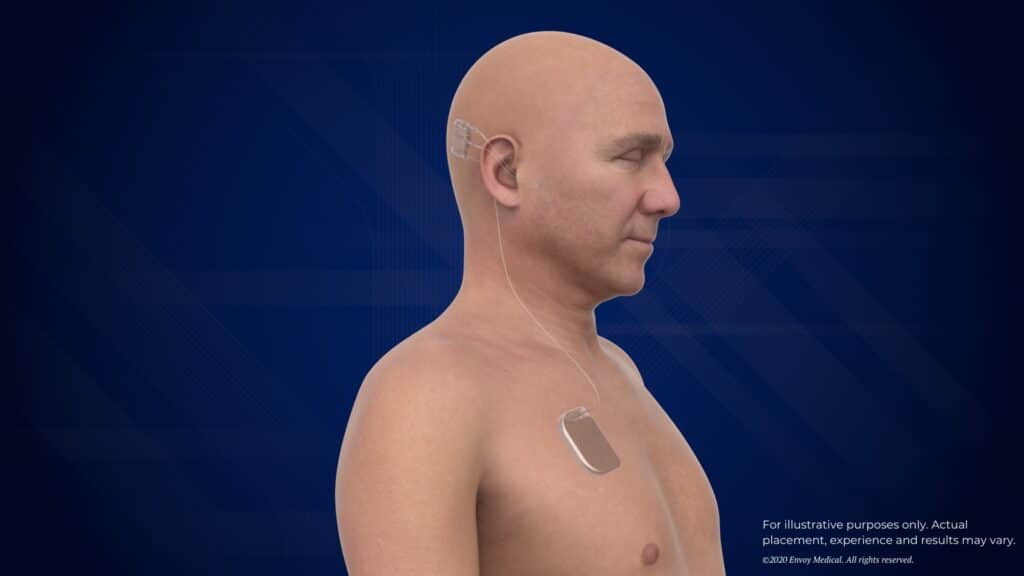
Envoy Medical’s Acclaim introduces a different architecture. CEO Brent Lucas outlines three key differentiators. The first is its sound pickup mechanism. Instead of an external or subcutaneous microphone, the Acclaim uses a piezoelectric sensor placed near the incus to capture the natural vibrations of the middle ear. “We leverage that natural organ by picking up the vibrations off the incus,” Lucas explains. “The way our device is designed is to have this natural sound coming in naturally as it was designed.”
The second difference is the absence of a magnet, which is typically used in traditional CIs to hold the external processor in place. The third is the power source: the Acclaim’s battery is implanted in the chest, connected to the processor via a lead. “Our battery is designed to last right now for several days between recharges,” Lucas says. “And then that battery will last probably 10 to 15 years before it needs to be replaced, and then it can be replaced in a very simple procedure, similar to how you might replace the battery of a pacemaker.”
Clinical Considerations and Candidacy
Currently, all completely implanted CIs in development are intended for adults with severe-to-profound sensorineural hearing loss, the same indication as traditional CIs. For Med-El’s TICI, the fitting range is identical to its traditional CI. “It’s more about the person’s individual needs,” says Robinson. “If the person’s need is freedom from an external device, then they don’t need to compromise on their expected hearing performance.”
Envoy’s use of a middle ear sensor means its device requires a healthy middle ear. “Anybody with an abnormal middle ear— a missing middle ear bone or a missing tympanic membrane, something like that—that could be a problem for this device,” Lucas notes.
Beyond audiometric criteria, patient lifestyle and capability are emerging as important factors. Arnedt suggests a fully implanted CI would function well for someone with an active lifestyle but also highlights another potential group. “I think that there is another subset of individuals that would greatly benefit from an internal fully implanted device, and that is the more elderly, maybe some who are dealing with some cognitive issues,” she says. For patients who find managing, maintaining, or keeping track of an external device overwhelming, a fully implanted solution could reduce patient and caregiver burden.
Regarding surgical risks, all manufacturers state that the risks are comparable to those of traditional cochlear implantation. The procedures involve a mastoidectomy and require general anesthesia. Lucas notes that Envoy’s system involves a second incision site for the chest-implanted battery, but anticipates any additional risk to be nominal.
The Road to Market
With clinical trials underway, a frequent question from both patients and professionals is when these devices will be available. Envoy Medical appears closest to market, with Lucas anticipating potential FDA approval by late 2027 or early 2028. “We are in a pivotal clinical study as we speak,” he says. And enrollment for it is now complete.
Cochlear and Med-El are more cautious with their timelines. “At this time, a commercial fully implantable cochlear implant is not available—all devices are for investigational use only,” says Janssen. “Further research, along with regulatory evaluation, will be required before this technology can be made accessible to the public.” Robinson of Med-El concurs, noting that with all companies in the investigational stage, it is too early to make definitive comparisons or timeline predictions.
This raises a critical question for hearing care professionals counseling patients today: should a candidate for a CI wait for a completely implantable model? Representatives from Cochlear and Med-El strongly advise against it. “People living with hearing loss today should not delay treatment or wait for future technologies,” Janssen states, a sentiment Robinson echoes. “Research has shown over and over again that the sooner hearing loss is treated adequately, the better the results,” she says.
Lucas offers a different perspective. While agreeing that those who are ready and need to address their hearing loss immediately should proceed with available options, he suggests some patients may have a choice. “Once you choose a cochlear implant, it’s really the idea that that’s your device for the rest of your life,” he says. “So, if you’re a borderline candidate or you want to wait a little bit, wait. I think it’s OK to wait a couple of years to see where the technology is going to be.”
Once fully implantable CIs are available, representatives for both Cochlear and Med-El anticipate them being just an additional option to add to their existing portfolio, with traditional CIs maintaining their popularity. “Fully implantable cochlear implants also may not be the solution of choice for all recipients,” Janssen says. And he notes that, “If approved, they may not be available in all markets.”
The Near Future
While many questions remain to be answered through ongoing clinical trials and future research, work on completely implantable cochlear implants is steadily advancing, promising to expand options and potentially create a new wave of CI candidates who will be able to benefit from this technology.
Featured image: Envoy Medical’s Acclaim is one of three completely implantable cochlear implants still in development. A hearing aid can be used with this type of device, as shown here. Photo: Envoy Medical