|
Jes Olsen, general manager, Oticon Medical |
William Demant Holding introduces Oticon Medical, a new business that focuses on the development and marketing of bone-anchored hearing solutions.
Bone-anchored hearing solutions benefit those with hearing losses that cannot be treated with traditional hearing aids, including hearing loss due to malfunctioning of the ear canal or middle ear (conductive or mixed hearing loss), and single-sided deafness.
Oticon Medical is the culmination of years of work by Oticon’s development organization to produce a comprehensive program consisting of sound processors, implants, fitting software, patient accessories, and special tools for surgeons.
"Our dedicated focus on development implies that on a continuous basis we allocate considerable resources to the development of entirely new business areas, as clearly illustrated by the establishment of Oticon Medical," says Niels Jacobsen, president and CEO of William Demant Holding. "The market for bone-anchored hearing solutions is a high-growth market with considerable unexploited growth potential, but also a market in which only few manufacturers have the technical skill to act. Due to the low market penetration thus far we look at this project as a long-term initiative and we look forward to being instrumental in growing the market to its full potential.”
The global market for bone-anchored implant systems is estimated at $60 million to $70 million, and has historically grown by more than 20% annually.
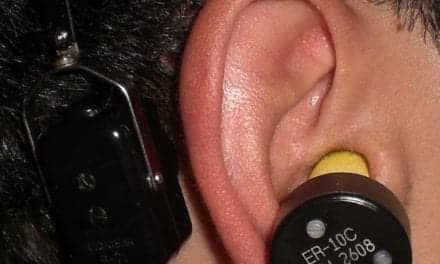
Approved by the FDA for conductive and mixed hearing loss in1996 and for single-sided deafness in 2002, bone-anchored implant systems feature an external sound processor held in place by an external abutment attached to a titanium screw that is surgically implanted behind the ear. The bone carries vibrations that bypass the external and middle ear and travel directly to the inner ear. An important component of the bone-anchored implant is the sound processor, which handles the actual sound processing.
Oticon Medical’s bone-anchored hearing solutions will feature sound processors based on Oticon’s proprietary RISE platform, an advanced technological platform. The company will emphasize products, software, and tools that deliver flexibility, safety, and patient outcomes and a higher rate of successful fittings to audiologists.
Until now, the market for bone-anchored hearing solutions has consisted of a few key markets including the United States, Scandinavia, the United Kingdom, and the Netherlands, where reimbursements are available for these treatment options, the company says. The company expects its entry into the market to continue to expand penetration of the existing markets and build new markets to increase patient access to the new technology.
“With a maturing therapy and increased acceptance of osseointegration, the time is right for a company with the dedicated focus and resources needed to better serve patients who cannot benefit from traditional hearing solutions,” says Jes Olsen, general manager of Oticon Medical. “Our goal is to introduce breakthrough concepts to meet this growing patient demand.”
Olsen, an Oticon A/S employee since 1986 and most recently VP of research and development, will drawn upon his strong audiological profile, his development experience within all competence areas, and his business expertise in the hearing aid industry.
Oticon Medical plans to tap the expertise of Oticon Medical staff members and of a range of resources within the William Demant Group, including Oticon’s 100-plus years of experience in audiology and sound processing and established manufacturing and logistics infrastructure. The company expects its strategic diversification of competencies and resources will help maximize its potential to develop breakthrough products.
“Our focus is patient-centered,” says Olsen. “We aim to provide the advanced technology solutions and the quality service, support, and training hearing care providers require to optimize successful outcomes for patients with conductive and mixed hearing loss and one-sided deafness.”
Since bone-anchored implant systems require patients to undergo a surgical procedure, Oticon Medical will work closely with leading otolaryngologists, neuro otologists, and clinics worldwide that specialize in these types of surgical procedures.
Oticon Medical plans to introduce the company’s first products in fall 2009.
[Source: Oticon]