Research | July 2016 Hearing Review
A study of 11 low-end and high-end hearing aids and PSAPs
This study evaluating 11 low-end and high-end hearing aids and PSAPs showed that all high-end hearing aids were able to fit most audiometric configurations, and two high-end PSAPs and one app were able to fit up to a moderate loss configuration. Most low-end hearing aids and PSAPs had inappropriately high amounts of low-frequency gain. High-end PSAPs provided appropriate levels of amplification and directional benefit for users but only for mild to moderate losses. Some low-end PSAPs and hearing aids were found to be inappropriate for any configuration and severity of high-frequency hearing losses.
Current estimates place the number of Americans age 50 or older with a measureable hearing loss at 26.7 million.1 Most forms of sensorineural hearing loss are not medically treatable, and without intervention can have a severe negative impact on an individual’s quality of life.2 Hearing loss has been positively correlated with increased likelihood of dementia,3 cognitive impairment,4 and social isolation.5 Hearing aids, along with aural rehabilitation and counseling, are considered the gold standard of care for those suffering from a hearing loss,6 yet hearing aid adoption rates have historically remained low, with just 14.2% of hearing aid candidates actually purchasing them.1
Recent research regarding the low rate of hearing aid adoption has examined the issues of affordability,7,8 device performance,8 and patient outcomes.9 While results have differed slightly between studies, it is apparent that the issue of affordability plays a significant role in the decision to purchase hearing aids.7,8 Obtaining hearing aids from a licensed hearing aid dispenser can cost upwards of $1,000 for two basic devices (in a bundled-service model) with costs rising in excess of $6,000 for the inclusion of more advanced features.
Confounding the affordability dilemma is the lack of standardization regarding third-party coverage of hearing aids, with many insurance companies—including traditional Medicare—refusing to pay for the devices.7 During periods of recession, hearing aid sales have been shown to dip significantly, indicating hearing healthcare may be seen as a luxury in times of economic duress.10
Given this information, it would seem logical for the hearing aid industry to attempt to reduce prices; however, hearing aid prices have previously shown to be fairly inelastic, with a reduction in prices not resulting in an increase in sales.11,12 Additionally, in countries where hearing aids are provided to patients at low or no cost, hearing aid adoption rates show only slight improvement over those in the United States.13,14
With passage of The Affordable Care Act in 2010, the US healthcare industry has paid special attention to increasing the efficiency and affordability of modern healthcare. In an effort to examine the role of federal regulations on the cost and accessibility of hearing healthcare, the Institute of Medicine (IOM) has undertaken a consensus study soliciting the opinions of providers, manufacturers, regulators, and researchers to determine what changes can be made to address the future needs of those with hearing loss. A division of the National Academies of Sciences, Engineering & Medicine, the IOM is a non-profit dedicated to providing objective analysis of scientific issues to influence and inform American public policy. While the results of this study will become available in June 2016, the need for affordable devices is already being addressed with some products in the market.
Efforts have been made to combat the affordability problem of hearing aids with a category of devices called personal sound amplification products, or PSAPs. These devices typically retail for thousands less than traditional hearing aids, and can be purchased directly by the consumer, without consultation from a licensed hearing aid provider. In October of 2015, the President’s Council of Advisors on Science and Technology (PCAST) released a report15 detailing recommendations for the future of hearing healthcare in the United States and the growing number of Americans experiencing hearing loss. Included in that report was the recommendation that “Americans would be better served if non-surgical air-conduction devices intended to address bilateral, gradual-onset, mild-to-moderate age-related hearing loss…were available over the counter.”15 While no true “over the counter” (OTC) hearing aid option currently exists, the PCAST has called on the FDA to change how it regulates both PSAPs and hearing aids in an attempt to make devices more affordable, encourage innovation, and reduce the stigma associated with hearing aid use.
For the average consumer, a hearing aid and a PSAP could appear identical. After all, the essential function of both types of devices is to amplify sound. However, according to an FDA draft guidance issued in 2013, a PSAP is “intended to amplify environmental sound for non-hearing impaired consumers.”16 The draft guidance adds that PSAPs should not be marketed as hearing aid replacements, nor should they be considered an OTC hearing aid. Thus, the definition of a hearing aid versus a PSAP relies on “intended use” and not on the function of the product.
As this is a non-legally-binding draft guidance published in 2013, PSAPs are not currently under regulation by the FDA. In contrast, hearing aids are categorized by the FDA as a Class I medical devices intended for use by those with hearing loss. Class I medical devices, while the lowest level of regulation afforded by the FDA, requires manufacturers to pay thousands of dollars in registration fees, register their products before commercial release, maintain several years of customer records, and most importantly requires companies to adhere to stringent manufacturing and design oversight. In their report,15 PCAST argues that this degree of regulation is unnecessary for hearing aids, as device failure is unlikely to result in injury to the end-user. Additionally, they argue this oversight has resulted in a stagnation of innovation in the hearing aid industry. As evidence, they point to the explosion of PSAPs in the consumer marketplace in just a few short years, having features like wireless communication and mobile phone compatibility that has taken much longer for the hearing aid industry to adopt.
Complicating matters is the recent introduction of a third class of amplification products called hearables. These devices, borrowing their name from the trend of wearable devices, are very similar to PSAPs. Most of the devices are wireless in-the-ear style headphones that combine the functionality of wireless headphones with additional features, including heart rate monitors, media storage, Bluetooth-compatibility, and equalizer settings allowing users to alter the sound quality of incoming audio signals. Some of these devices are not just intended to be worn when listening to music, but can be worn for extended periods of time to alter how we hear environmental sounds and monitor our surroundings. Technology consultant Nick Hunn projects that hearables, having only gained traction in the last couple of years, will become a $50 billion industry by 2018.17 With most hearables entering the market at around $500, these devices are almost identical to some high-end PSAPs, and their unregulated status may only further confuse the average consumer.
While groups like the American Speech-Language Hearing Association (ASHA) and the International Hearing Society (IHS) have expressed concern regarding the recommendations included in the PCAST report, the question remains: Are there PSAPs that could effectively act as an OTC hearing aid? Because of the inexpensive and relatively under-regulated nature of PSAPs, there is limited data about their real-world performance. Research by Cheng and McPherson18 in 2000 examined several PSAPs using real-ear measurements, and found the devices often provided too much low frequency gain and insufficient high frequency gain. This configuration made the devices inappropriate for many users with a high frequency hearing loss. A 2008 study by Callaway and Punch19 obtained similar results across a range of PSAPs, but did find that higher-end PSAP models (those above $300) were able to more closely match NAL-NL1 prescriptive targets and appropriately amplify sound. Subjectively, patients have reported no discernable difference between environmental sounds and music amplified through a mid-range PSAP and a traditional BTE hearing aid. However, there was significant preference for speech amplified through the hearing aid versus the PSAP.20 These studies suggest that PSAPs may be appropriately fit to some types of hearing losses, but further data is required before that assumption can be made.
In summary, PSAPs encompass a wide range of devices, some of which may be suitable for those who cannot afford a hearing aid or are seeking a low-cost introduction to amplification. The purpose of the present study was to measure the objective benefit of a range of PSAPs and determine their appropriateness as amplification devices for several common audiometric configurations. In Part 1 of the study, 7 PSAPs and 4 low- and high-end hearing aids were electroacoustically analyzed in an Aurical Hearing Instrument Test (HIT) box to determine output characteristics of the device under controlled conditions, with the resulting data compared to standards put forth by the ANSI 2009 standards for hearing aids.21 In Part 2, each device was monaurally fit on a KEMAR mannequin and real-ear measurements obtained to NAL-NL2 prescriptive targets.22
Hearing Devices Tested
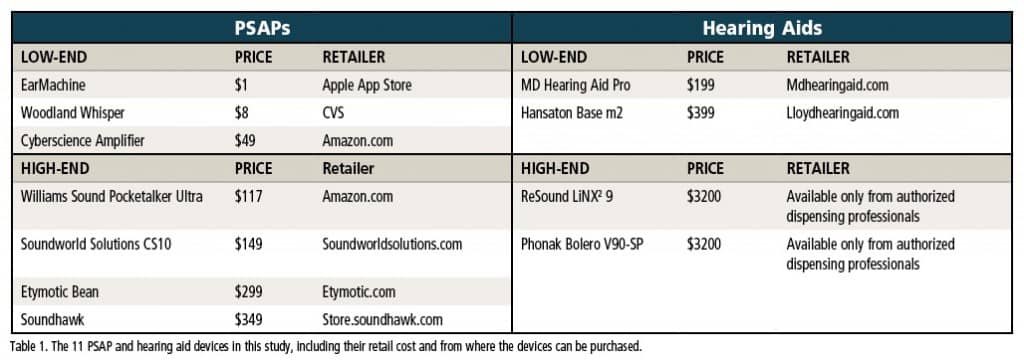
Of the PSAPs, one was an iPhone-based application (EarMachine), one was a body-worn device (Pocketalker), three were in-the-ear style devices (Etymotic Bean, Woodland Whisper and Soundhawk), and two were behind-the-ear styles (Cyberscience Amplifier and Soundworld Solutions CS10). All the hearing aids were behind-the-ear style (BTE) aids (MD Hearing Aid Pro, Hansaton Base m2, ReSound LiNX2 9, and Phonak Bolero V90-SP). Of additional interest, all devices included in this study, excluding the high-end hearing aids, can be purchased directly by the consumer without consulting a hearing care professional.
Procedures
As noted previously, the study was comprised of two phases: 1) Tests within the Hearing Instrument Test (HIT) box, and 2) Tests involving on-ear fitting capabilities.
Phase 1. Each device was coupled to a 2-cc coupler within a GN Otometrics Aurical Hearing Instrument Test (HIT) chamber and underwent a complete electroacoustic analysis according to ANSI S3.22 standards, including Maximum Output Sound Pressure Level (OSPL90), Equivalent Input Noise (EIN), Total Harmonic Distortion (THD), and Directionality:
- Comparison of the high-frequency average OSPL90 to the OSPL90 at 500 Hz allows determination of the ratio of low-frequency to high-frequency gain. Prior research indicates many low-end devices deliver too much low-frequency gain for the typical presbycusis audiometric configuration.
- Equivalent Input Noise (EIN) is a measure of the noise generated by the internal components of the device, as measured in decibels.
- Total Harmonic Distortion (THD) indicates the degree to which the selected devices introduce additional unwanted harmonics into the amplified signal.
- Directionality is the difference in output between the sound measured from the front of the device compared to from the rear.

Due to size and equipment limitations, the EarMachine application was tested in an Audioscan Verifit test chamber rather than the Aurical. EarMachine was loaded onto an Apple iPhone 6 Plus using standard Apple “earpod” earbud-style headphones. These headphones are included with the purchase of the phone and, as such, represent the most likely headset a consumer would use with the EarMachine product. In total, all measurements were repeated 10 times, with the device being uncoupled and recoupled with each replication, and with no two measurements repeated in the same day.
Phase 2. The second phase of the study examined the on-ear fitting capabilities of each device. A total of 10 audiograms were entered into the Noah Software suite. Using the computerized GN Otometrics real-ear software module, a KEMAR mannequin was placed at a distance of 2 feet and facing (0° azimuth) the Aurical real-ear speaker. The system was calibrated before each round of tests. A probe-tube microphone was placed within the right ear cavity, and unoccluded insertion gain was measured with no device in the ear. Following that measurement, the device was placed in the right ear of the mannequin and aided output was measured using the International Speech Test Signal (ISTS) presented at 50 dB (soft), 65 dB (average), and 80 dB (loud) intensities. The output of the device was then adjusted to match as closely as possible the NAL-NL2 targets generated by the real-ear software. This process was repeated 10 times, with no more than one round of measurements taken on a given day.
To determine if the selected devices were appropriate for a given patient population, 10 audiometric configurations were chosen for this study (Table 3). Ranging from a very mild hearing loss to a profound hearing loss, and split between flat/moderately sloping and precipitously sloping, these 10 audiograms were designed to represent the entire range of high-frequency hearing losses encountered in a clinical setting, based on 28,244 audiograms.23 Because age-related hearing losses are typically high-frequency in nature, this set of audiograms represent the range of hearing configurations of a traditional adult hearing aid or PSAP consumer.
Based on prior clinical research by Punch, a target is considered matched if the achieved amplification is within 10 dB of the NAL-NL2 target at a given frequency (250, 500, 750, 1,000, 1,500, 2,000, 3,000, 4,000, & 6,000 Hz). At each intensity level, one target can be missed and still be considered “fit to NAL-NL2 targets.” The fitting percentage is determined by dividing the total number of achieved targets by the 27 total NAL-NL2 targets. With an allowance of 1 missed target per intensity, a fitting score of 88% or above was considered an “appropriate fit” for that audiometric configuration. NAL-NL2 targets were selected for this particular study due to their focus on audibility and speech understanding in addition to prior research indicating a preference for NAL-NL2 over other prescriptive methods.24 (In a clinical setting, best practices indicate the use of any research-backed fitting formula.)
Results
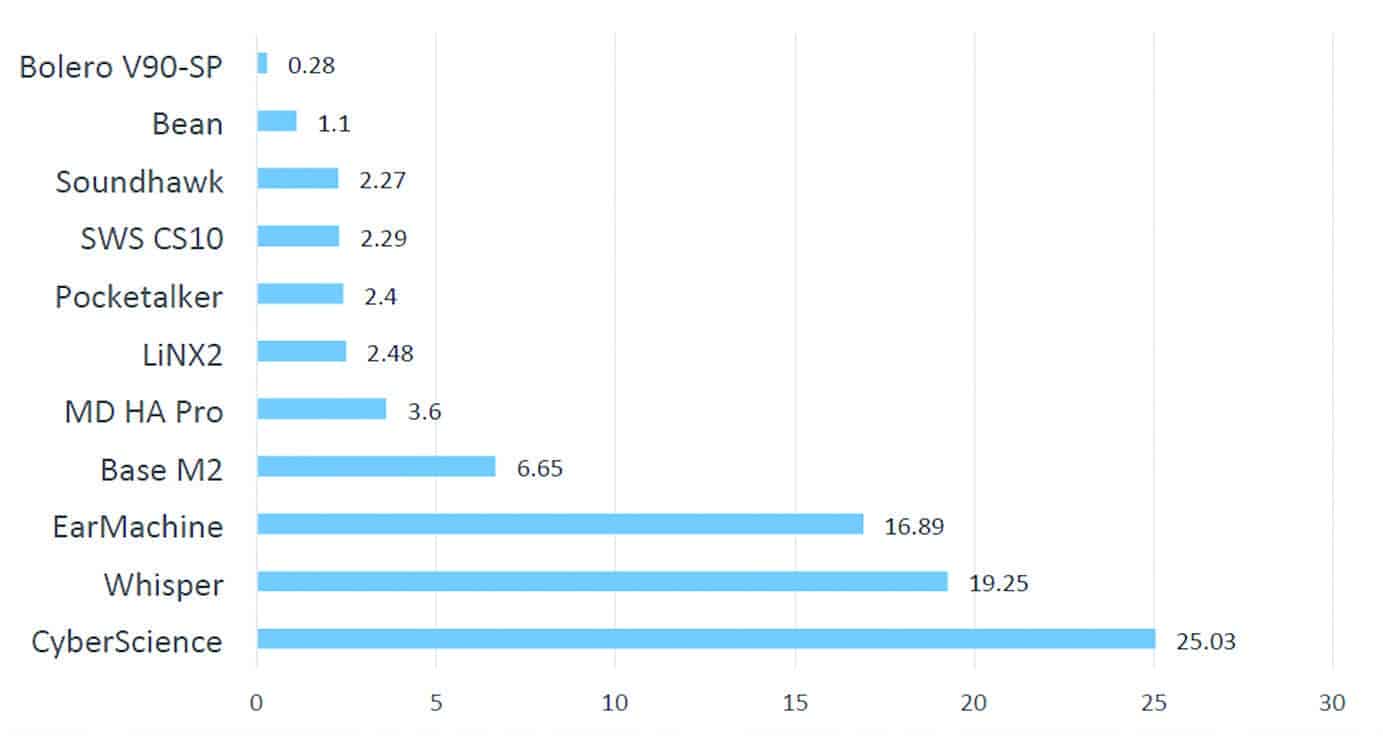
Differences in low- and high-frequency output (OSPL90). Figure 1 shows the average difference in output between the OSPL90 HFA and the OSPL90 value at 500 Hz in decibels. A smaller difference is indicative of similar output characteristics between the low frequency and high frequency outputs, while a larger difference indicates a sizable imbalance between low- and high-frequency outputs. The worst performers in this category were the low-end PSAPs, with disproportionately high levels of low frequency gain and little high frequency gain. The remaining devices demonstrated proportionate levels of high- and low-frequency gain.
Internal noise. Figure 2 shows the average level of equivalent input noise for each device. Of the 11 devices tested, only 4 were less than the ANSI standard of 28 dB SPL. Overall, the low-end PSAPs performed the worst in this test, featuring high amounts of internal noise. All devices that tested within ANSI specifications for noise were classified as hearing aids.
A high degree of input noise is not necessarily inappropriate for a given patient. If their unaided thresholds are above the level of the input noise, then that patient will not hear the noise generated by the device. This should be taken into consideration on a case-by-case basis.
Harmonic distortion. Figure 3 shows the average total harmonic distortion for each device as measured at 500, 800, and 1,600 Hz. Overall, most devices were well within the suggested 3% tolerance, suggesting even the low-cost devices amplify with little added distortion to the target signal. Only the MD Hearing Aid Pro and the Williams Sound Pocketalker Ultra introduced more than 3% total harmonic distortion at any one frequency tested.
Directional benefit. Figure 4 shows the average directional benefit of each device. According to Killion,25 a signal-to-noise ratio (SNR) of 2 dB or greater is needed for a noticeable directional benefit. Of the 11 devices, only three had a directional benefit above 2 dB, two of them being the high-end hearing aids.
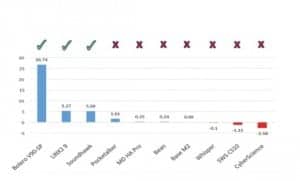
Figure 4. “Average directionality” results using the Audioscan Aurical system. It should be noted this test is not intended to be a measure of real-world directivity (ie, such tests can only be performed in a soundfield), but instead represents a binary (yes/no) test for the presence of directionality, where perceptible benefit via directionality is thought to begin somewhere above 2 dB, denoted here by green checks (see Killion25). [See clarification at end of article.]
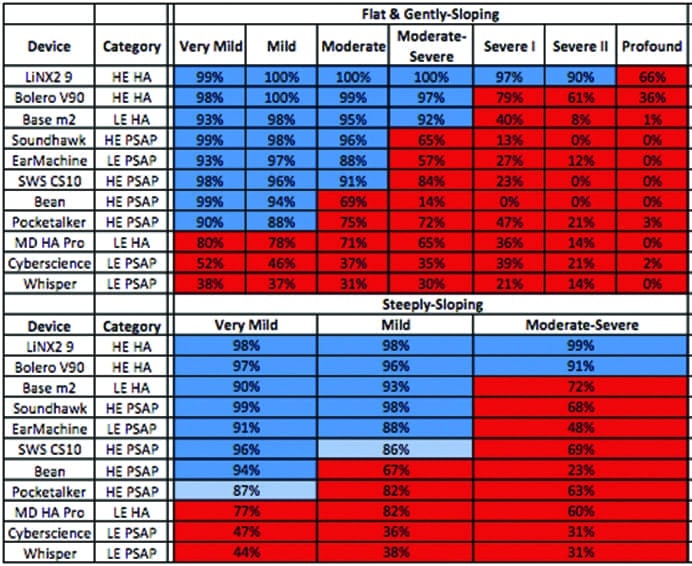
Matching NAL-NL2 fitting targets. Table 4 shows the average percentage of NAL-NL2 targets met by each device for each audiometric configuration. The high-end hearing aids were able to appropriately match targets for the most hearing loss configurations of all devices, with the ReSound LiNX2 9 fitting 9 of 10 audiograms and the Phonak Bolero V90-SP fitting 7 of 10 audiograms. Following the high-end hearing aids, the best performer was the Hansaton Base m2 low-end hearing aid, which was able to match targets up to a moderately-severe hearing loss, or 6 of 10 audiometric configurations.
The best performing PSAP, the Soundhawk, was able to appropriately match targets up to a moderate hearing loss, or 5 of 10 audiometric configurations. Of the remaining high-end PSAPs, each was able to match at least a mild hearing loss, or 3 of 10 audiometric configurations.
Three of the 11 devices (MD Hearing Aid Pro, the Cyberscience Amplifier, and the Woodland Whisper) were unable to adequately meet targets for any hearing loss and, as such, were deemed by this study to be inappropriate for all the audiometric configurations tested.
Discussion
The variability shown in the results of this study illustrates the problematic nature of FDA regulation of the hearing aid industry. Some devices, like the MD Hearing Aid Pro, are subjected to full FDA regulations and are classified as a hearing aid, yet they are outperformed by a high-end PSAP. While FDA regulation is intended to ensure device quality and patient safety, that effect is not entirely evident in the results of this study.
With this is mind, is FDA regulation serving a beneficial purpose at this point? For patients with a very mild, mild, or moderate degree of hearing loss, some high-quality PSAPs could offer a cost-effective introduction to amplification compared to traditional FDA-regulated hearing aids. Additionally, if a patient loses or damages his or her hearing aids, these devices might offer temporary assistance until a replacement hearing aid can be obtained (should it be proven that these PSAPs do not detract from real-world performance in everyday listening situations).
Having shown that high-end PSAPs can be appropriately fit to NAL-NL2 amplification targets for a limited range of hearing losses, further research should be done with human participants to determine real-world device performance. Although the results of this study are promising, high-end hearing aids include programming options and acoustic processing strategies, such as wide dynamic range compression (WDRC), feedback suppression, and directional microphones designed to enhance the comfort and quality of the device output. These features were often lacking in the PSAPs tested in this study.
While electroacoustic analysis can measure the objective quality of the output, there are many factors that can impact a patient’s overall satisfaction. These factors reach far beyond sound quality and include such things as cosmetic concerns, assistive listening device compatibility, device reliability, and the involvement of a skilled hearing healthcare provider. However, research conducted by Sergei Kochkin comparing traditional hearing aid patient satisfaction rates to those who purchased direct-mail hearing aids showed that consumers are willing to make trade-offs in benefit for substantial reductions in price.26 The direct-mail hearing aids in Kochkin’s study delivered “about average” real-world benefit but significantly less benefit than those hearing aids dispensed by professionals who had adopted the highest levels of evidence-based best practices.
This line of research could have profound implications on the dispensing of hearing aids in the future. Many hearing aid provider practices rely on income generated from the sale of hearing aids to support personnel and clinic expenses. With the introduction of low-cost devices, some hearing aid providers may be reticent to adopt the technology fearing profit losses. High-quality PSAPs, should they be made available to those with hearing loss, have the ability to revolutionize the industry and potentially expand the number of hearing-impaired patients who have access to economical hearing care, particularly for those consumers currently underserved by existing technologies.
Regardless of technology, this research demonstrates the importance of performing independent verification measures on all amplification devices. As our testing demonstrated, there is tremendous variability in quality between devices, with those regulated as hearing aids not necessarily representing a higher-quality device than those not FDA-classified as hearing aids. For those unaccustomed to performing these tests, the preceding measures are some things to begin looking at when performing an electroacoustic analysis. As for real-ear measurements, best practices indicate we should be completing these measures during every fitting to ensure patients receive the appropriate level of amplification using evidence-based fitting formulas.
Summary and Key Findings
This study examined differences in output between 11 high-end and low-end personal sound amplification products (PSAPs) and hearing aids, and determined the high-frequency hearing losses for which these devices provide appropriate gain and output. The 11 devices in this study were divided into two groups: those classified by the FDA as a hearing aid, and those classified as a PSAP. These two groups were further divided into low-end and high-end categories, based on retail price at time of publication. Each device was electroacoustically evaluated according to ANSI S3.22 standards in a hearing instrument test chamber and the results were compared across devices. Additionally, gain targets from NAL-NL2 were calculated for 10 different audiometric configurations using KEMAR and the number of targets matched was averaged over 10 trials for a total fitting score out of 100.
The results indicated:
- High-end hearing aids were able to meet NAL-NL2 targets for the most audiometric configurations. Two high-end PSAPs and one app were able to meet NAL-NL2 targets up to a moderate high frequency hearing loss.
- Most low-end hearing aids and PSAPs had inappropriately high amounts of low frequency gain.
- Directional benefit was present in only the two high-end hearing aids and one of the high-end PSAPs.
- Overall, most devices included in this study had a high degree of internal noise, with only devices in the hearing aid category achieving ANSI standards.
- High-end PSAPs provided appropriate levels of amplification and directional benefit for users with high-frequency hearing losses ranging from mild to moderate in severity.
- Some low-end PSAPs and low-end hearing aids are inappropriate for any configuration and severity of high-frequency hearing losses.
Acknowledgements
The lead author (CS) thanks everyone on his committee for their time and input on this project. The authors also thank Drew Dundas, PhD, of Soundhawk for the use of their device during this project. Additionally, we thank Amyn Amlani, PhD, of the University of North Texas for his input during the project design and with device troubleshooting, as well as Brian Taylor, AuD, and Pamela Souza, PhD.
Clarification
The authors would like to clarify the results of the electroacoustic directionality testing (Figure 4) in our study. Electroacoustic directionality testing completed in the Aurical testbox is a binary test designed to indicate the presence or absence of a directional advantage with broadband noise presented at the front of the device versus the rear. As such, it is not a direct measure of real-world directional microphone performance or benefit and should not be interpreted as such. For the purposes of this study, any device with a directional average of 2 dB or greater was considered to have directional functionality.
In an effort to further explain the results in Figure 4, a column has been added to the top of the figure that indicates whether or not the specified device has met the 2 dB threshold for directional functionality in the test box. Of the 4 devices in this study with directional microphone technology, only 3 of them had noticeable directionality: both of the high-end hearing aids and one high-end PSAP. Although it appears the Phonak Bolero greatly outscored the other two devices, it is uncertain what real-world performance with speech stimuli would be for any of these devices.
Also see: Letter: On ‘PSAPS vs Hearing Aids’ and the MDHearingAid Analog Device
References
-
Chien W, Lin F. Prevalence of hearing aid use among older adults in the united states. Arch Intern Med. 2012;172(3):292.
-
Kochkin S. MarkeTrak VIII: Patients report improved quality of life with hearing aid usage. Hear Jour. 2011;64(6):25-32.
-
Gurgel RK, Ward PD, Schwartz S, Norton MC, Foster NL, Tschanz J. Relationship of hearing loss and dementia: A prospective, population-based study. Otol Neurol. 2014;35:775-781.
-
Chisolm TH1, Johnson CE, Danhauer JL, Portz LJ, Abrams HB, Lesner S, McCarthy PA, Newman CW. A systematic review of health-related quality of life and hearing aids: Final report of the American Academy of Audiology task force on health-related quality of life benefits of amplification in adults. J Am Acad Audiol. 2007;18:151-183.
-
Cacioppo JT, Hawkley LC, Norman GJ, Bernston GG. Social isolation. Ann NY Acad Sci. 2011;1231[Aug]:17-22. doi: 10.1111/j.1749-6632.2011.06028.x
-
Valente M, Abrams H, Benson D, Chisholm TH, Citron D, Hampton D, Loavenbruck A, Ricketts T, Solodar H, Sweetow R. Guidelines for the audiologic management of adult hearing impairment. Am Acad Audiol. 2012. Available at: http://audiology.com/haguidelines%202007.pdf
-
Bainbridge K, Ramachandran V. Hearing aid use among older U.S. adults: The national health and nutrition examination survey, 2005-2006 and 2009-2010. Ear Hear. 2014;35(3):289-294.
-
Kochkin S. MarkeTrak VIII: The key influencing factors in hearing aid purchase intent. Hearing Review. 2012;19(3):12-25. Available at: https://hearingreview.com/2012/03/marketrak-viii-the-key-influencing-factors-in-hearing-aid-purchase-intent
-
Halpin C, Rauch S. Improvement in word recognition score with level is associated with hearing aid ownership among patients with hearing loss. Audiol Neurol. 2012;17:139-147. doi: 10.1159/000333777
-
Amlani A, De Silva D. Effects of economy and FDA intervention on the hearing aid industry. Am J Audiol. 2005;14:71-79.
-
Lee K, Lotz P. Noise and silence in the hearing instrument industry [working paper]. Frederiksberg, Denmark: Dept Industrial Economics & Design, Copenhagen Business School;1998.
-
Amlani A. How patient demand impacts pricing and revenue. Hearing Review. 2008;15(3):16-19. Available at: https://hearingreview.com/2008/03/how-patient-demand-impacts-pricing-and-revenue
-
Hougard S, Ruf S. EuroTrak I: A consumer survey about hearing aids in Germany, France, and the UK. Hearing Review. 2011;18:23-28. Available at: https://hearingreview.com/2011/02/eurotrak-i-a-consumer-survey-about-hearing-aids-in-germany-france-and-the-uk
-
Godinho L. What Is the Most Efficient Reimbursement System in Europe? A statistical look at the wide range of European hearing aid reimbursement systems. Hearing Review. 2016;23(1):16. Available at: https://hearingreview.com/2015/12/efficient-reimbursement-system-europe
-
President’s Council of Advisors on Science & Technology (PCAST). Aging America & hearing loss: Imperative of improved hearing technologies. October 2015. Available at: https://www.whitehouse.gov/sites/default/files/microsites/ostp/PCAST/pcast_hearing_tech_letterreport_final.pdf
-
US Food and Drug Administration (FDA). Regulatory Requirements for Hearing Aid Devices and Personal Sound Amplification Products–Draft Guidance for Industry and Food and Drug Administration Staff. Washington, DC: FDA. Nov 7, 2013. Available at: http://www.fda.gov/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/ucm373461.htm
-
Hunn N. The Market for Smart Wearable Technology: A Consumer Centric Approach. WiFore Consulting. February 2015. Available at: http://www.nickhunn.com/wp-content/uploads/downloads/2014/08/The-Market-for-Smart-Wearables.pdf
-
Cheng CM, McPherson B. Over-the-counter hearing aids: Electroacoustic characteristics and possible target client groups. Audiology. 2000;39:110-116.
-
Callaway S, Punch J. An electroacoustic analysis of over-the-counter hearing aids. Am J Audiol. 2008;17:14-24.
-
Breitbart D. Is there a preference between hearing aids and personal sound amplification products (PSAPs)? AuD research project, University of Memphis;April 2014. Available at: http://www.harlmemphis.org/files/2513/9628/6861/Breitbart2014.pdf
-
American National Standards Institute (ANSI). ANSI S3.22-2009, American National Standard Specification of Hearing Aid Characteristics. New York: ANSI; 2009.
-
Keidser G, Dillon H, Flax M, Ching T, Brewer S. The NAL-NL2 prescription procedure. Audiol Res. 2011;1(e24):88-90.
-
Bisgaard N, Vlaming S, Dahlquist M. Standard audiograms for the IEC 60118-5 measurement procedure. Trends Amplif. 2010;14(2):113-120.
-
Johnson EE. Modern prescription theory and application: realistic expectations for speech recognition with hearing aids. Trends Amplif. 2013;17[Mar/Apr]:143-170. doi: 10.1177/1084713813506301
-
Killion M. Myths about hearing in noise and directional microphones. Hearing Review. 2004;11(2):14-19,72,73. Available at: https://hearingreview.com/2004/02/myths-about-hearing-in-noise-and-directional-microphones
-
Kochkin S. A comparison of consumer satisfaction, subjective benefit and quality of life changes associated with traditional and direct-mail hearing aid use. Hearing Review. 2014;21(1)[Jan]:16-26. Available at: https://hearingreview.com/2014/01/a-comparison-of-consumer-satisfaction-subjective-benefit-and-quality-of-life-changes-associated-with-traditional-and-direct-mail-hearing-aid-use
Correspondence can be addressed to HR or Chase Smith at: [email protected]
Original citation for this article: Smith C, Wilber LA, Cavitt K. PSAPs vs Hearing Aids: An Electroacoustic Analysis of Performance and Fitting Capabilities. Hearing Review. 2016;23(7):18.?








Excellent Article and a testament to using Real Ear or test box verification in your fittings
With a PSAP over the counter or ordering Via email. how does one address a medical referral, medical problems, medical issues related to their problem? Regular physical exams will not disclose most if not all of those issues.
As a practitioner I have had about 5 to 10 medical referrals a month. with approximately 40 to 50 patients a month That means those people will not have the proper attention to their real issue. The bigger concern is how many people will purchase assisted listening devices instead of having proper medical attention for their problem?
Are there any statistics with some ratio of events of medical needs related to patient seeking Assisted listening devices?
Excellent article on a truly difficult subject. As someone just finding a need for improved hearing, I have struggled to make a decision. My main concern is purchasing an expensive hearing device and either not using it, breaking it, or loosing it. I think a good PSAP would be a good first choice but I understand the Soundhawk Co. is no longer in business. Is there a good 2nd choice PSAP or something coming worth waiting for?
Perhaps outside the scope of this research, but have recent developments dealt with tinnitus? As a drummer for 40 years (very loud hard rock in the 70’s and 80’s) I suffer from dips at 5K and above 11K as well as severe tinnitus.
Thanks for a great article!
Well researched but with a major flaw: You never match the hearing aid to an audiogram. You match the hearing aid to the cognitive representations of that specific hearing loss.
It is obvious that PSAPs lack the complexities necessary to improve on cognitive deficits which are end points of any type of hearing loss.
Please delete this article from your archives at it is misleading to consumers and professionals alike.
Jay Muhury.
While I would certainly agree that an audiogram is only the starting point for fitting a patient, I do not think there is evidence that “it is obvious that PSAPs lack the complexities necessary to improve on cognitive deficits…” (in fact, definitive proof for hearing aids improving cognition is still out–but it’s looking very good). However, you CAN measure the performance of an amplification device to see how well it matches the acoustic needs of a patient (audibility and audition), which the authors attempt to do in this article with known, agreed-upon electroacoustic parameters. There is certainly a lot more research to be done in this area, and as I say in my editorial in this issue, this article poses almost as many questions as it does answers about the performance of PSAPs. As the authors state, the bottom line is hearing care professionals would be wise to verify (preferably, first in a HIT box) any new devices they are considering to recommend for their patients.
Who do you think you are telling such distinguished researchers to delete this article. What have YOU ever done regarding this? Do you think everyone is stupid that they don’t know that every contingency cannot be accounted for? Make your comment about what you think is lacking and keep your recommendations to yourself. Delete YOUR post.
I recommend reviewing table 3 “Audiometric Configurations”.