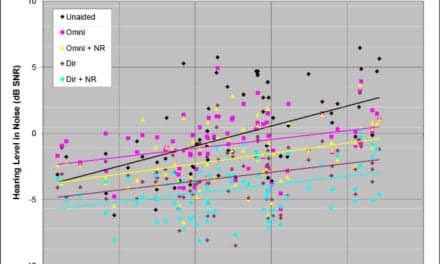
Cochlear Limited, a global provider of implantable hearing solutions, has announced the US Food and Drug Administration (FDA) approved Nucleus cochlear implants for magnetic resonance imaging (MRI) up to 3.0T with the magnet removed. Additionally, with the use of the Cochlear Nucleus Implant Bandage and Splint Kit, Nucleus implants are approved for 1.5T MRI with the magnet in place.
An MRI is a diagnostic tool to obtain images of organs and tissues using a very powerful magnetic field measured in tesla (T). MRIs can range in strength from 0.2T to 7.0T, with 1.5T being the most common. Cochlear Limited explains that inside each cochlear implant is a magnet, and nearly 20 years ago Cochlear introduced a key safety feature to ensure MRI compatibility for its implants: a removable magnet. The removable magnet ensures customer safety and comfort if an MRI is needed and an alternative imaging option is not available. The magnet is easy to remove and replace with a minor procedure if needed.

“Cochlear has always taken the lead in adapting our implants to MRI technology to ensure customer safety and comfort, as well as protecting the efficacy of the implant products that our customers depend on to hear,” said Tony Manna, president of Cochlear North America. “We are proud to be able to provide a range of MRI options–including the removable magnet and the splint kit–for our customers and their medical professionals to ensure safety, efficiency and convenience for all.”
If a 1.5T MRI with the magnet in place is needed, the Cochlear Nucleus Implant Bandage and Splint Kit can be ordered by clinics and hospitals, individual radiology clinics, and Cochlear customers. The kits are provided at no charge when ordered to minimize any financial burden to Cochlear customers and medical professionals.

According to the National Institute on Deafness and Other Communication Disorders, roughly 58,000 adults and 38,000 children have received cochlear implants in the United States.3 Nearly two million Americans could be candidates for cochlear implant technology, but only 5% of patients who can benefit have been treated. Cochlear Limited reports that it has helped over 450,000 people of all ages, across more than 100 countries, to hear through implant technology from Cochlear.
Source: Cochlear Limited; Cochlear Americas



Today I had to have an MRI of my head and neck at 1.5T to confirm a prolapsed disc in my neck prior to surgery.
It took weeks of consultation and uncertainties between the MRI clinic, my Cochlear Implant surgeon and my Neurosurgeon, mainly because nobody was willing to take my magnets out prior to the scan.
Anyhow, so today, the MRI techs seemed sceptical and stated that they’d never had anyone make it fully into the tunnel due to their pain being unbearable.
Anyhow, I made it into the tunnel, yes it hurt like hell, my head felt like a pumpkin and I rode it out for around ten minutes or so until I started to hear some rather unnerving feedback sounds from the elecrodes in both my ears so I knew that wasn’t good, to me that was suggestive of possible shifting of the implant. So that’s when I finally pulled the pin.
After my head was unbandaged I realised half the pain was from the splint kit supplied by cochlear, basically it is just two stiff rectangular pieces of acrylic-like card. The shape did not in any way conform to my implant curvature and it was hard as heck pressed up against them. If a better designed splint kit were available that was a snug fit and provided better cushioning it may improve MRI outcomes.
I’m one of the lucky ones that made it through the MRI, I got my diagnosis and I’d do it again if I had to.
I have Meniere’s disease it is a disorder of the inner ear that causes episodes in which you feel as if you’re spinning (vertigo), and you have fluctuating hearing loss with a progressive, ultimately permanent loss of hearing, ringing in the ear (tinnitus), and sometimes a feeling of fullness or pressure in your ear. My doctor said that he will be putting Nucleus CI512 in since I have a nerve issue not a bone issues. Does anyone have this implant, if so how do you like it?
Why can’t med el share their magnet technology with cochlear?
It will benefit the whole world to have all cochlear brands become compatible with the MRi instead of just one
Working together and sharing technical knowledge regarding implants benefits all of mankind.
Hello, my name is Berivan. I am 24 years old from Iraq. I need a contact for surgery because in 2001 in Baghdad on first surgery losed. Now I’m not good. My English not good sorry.
Hi Berivan
Can you send an enquiry to http://www.cochlear.com perhaps they can suggest a clinic in Iraq for you?
Good luck!
I waited for this kit for 4 months. It was released and overnighted to my doctor’s office Wednesday the 31st. I had my MRI on Thursday and after 2 minutes in the machine with the kit properly put on, I had I be pulled out because of the pain. It popped out of the Cochlear and I had to have surgery 2 hours later to remove the magnet. Sorry guys, but back to the drawing board.
Tiffany Gonzalez