Features | January 2015 Hearing Review
A brief review of important findings in the hearing healthcare literature
By Douglas L. Beck, AuD; Michelle Petrak, PhD; Jane R. Madell, PhD; and Sharon L. Cushing, MD
In 2015, children with vestibular and hearing problems need to be screened, diagnosed, and treated as quickly as possible to maximize successful habilitation. Professionals associated with pediatric hearing healthcare have addressed these issues for decades and the status quo today is vastly improved from 20 years ago.
However, pediatric vestibular and hearing disorders are not “mainstream” topics, and as such, there is still much to learn and incorporate into our clinical protocols. This article will review “state-of-the-art” ideas, concepts, and protocols with regard to pediatric vestibular and hearing disorders.
Overview of Pediatric Vestibular Disorders
Beck, Petrak, and Smith1 reported hearing care professionals (HCPs) often use multiple terms to describe essentially the same (or highly similar) phenomena, specifically with regard to vestibular complaints and disorders. Overlapping and non-exclusive test results can be confusing and are often interpreted with respect to the HCP’s background and knowledge.
However, the outcomes-based statistical strength of the individual test to accurately diagnose the specific disorder is often unknown. For example, migraine associated dizziness, vestibular migraine, benign recurrent vertigo, migraine related vertigo, migraine related dizziness, migraine associated vertigo, migraine associated dizziness, migraine related vestibulopathy, vertiginous migraine, and migrainous vertigo (and more) appear to share a common pathogenesis and similar test results, and arguably, each term may be applied to the same set of symptoms.
Casselbrant2 stated the most common vestibular disorder in children with normal otoscopic findings is vestibular migraine (VM). Of course, children do not have the knowledge or vocabulary to describe their symptoms in detail. Nonetheless, it is believed VM occurs without aura and is more common in females. It is often associated with headache, and can include nausea, vomiting, and photophobia, and may be related to menstrual periods. Casselbrant notes children may report motion sickness and/or carsickness. Nandi and Luxon3 report the most common etiologies of vestibular disorders in children include migraine, otitis media, and trauma. Others report up to 10% of vestibular issues in children may be due to visual problems/disorders.
Beck, Petrak, and Bahner4 reported vestibular disorders in children are under-diagnosed, and children with sensorineural hearing loss (SNHL) are at substantially greater risk for vestibular disorders than their age-related peers without SNHL. Nandi and Luxon3 stated roughly one-third of deaf people have unusual vestibular systems. Cushing and colleagues5 reported vestibular end-organ dysfunction was present in half their subjects with profound SNHL.
Overview of Pediatric Hearing Loss
The Joint Committee on Infant Hearing6 reports 1.4% of children who fail newborn hearing screenings have permanent hearing loss and 60% of infants who fail screening do not return for follow-up. The American Speech-Language-Hearing Association7 reports 3% of newborns have permanent hearing loss at birth and that number increases to 6% by the time children reach school age. The percent of children with unilateral hearing loss is estimated to be 6.3%.8 Joint Commission Guidelines9 suggest infants should be screened by 1 month of age, receive diagnostic services by 3 months of age, and receive intervention by 6 months of age.
Limb et al10 reports a significant number of children with permanent hearing loss do not receive hearing aids. A survey of state early intervention programs reported finances remain a significant factor for children who do not receive hearing aids.
Screenings vs Diagnostic Testing
Of note, children with normal peripheral hearing may have listening problems, such as auditory processing disorders (APD) and many (perhaps 15% of all children with SNHL) may have dys-synchrony disorders, such as auditory neuropathy spectrum disorder (ANSD). Neither of these listening disorders will be identified when otoacoustic emissions (OAEs) or pure-tone screenings are used in isolation. Further, for children with cytomegalovirus (CMV) and other anomalies, they may be born with normal hearing and may pass the screening, while substantial hearing loss may develop over the ensuing months and years.
That is, screenings are just that: screenings. Screenings generally identify hearing disorders present at that moment in time. Screenings do not guarantee or predict hearing or listening outcomes for specific children over the long-term.
Infant Hearing Evaluation
Newborn hearing screening identifies children who require further evaluation at the moment of their screening. Initial screenings are conducted using ABR and/or ASSR, tympanometry, and OAEs. Children who test positive (ie, are identified with probable hearing loss) receive ongoing behavioral hearing testing and are fit with hearing aids or other treatments as indicated.
Nonetheless, behavioral testing is critical because it provides information about how children respond to auditory information that is not available from electrophysiologic testing.11 Behavioral testing can be used to measure responses for infants. From birth to 6 months of age, behavioral observation testing utilizing sucking responses is appropriate,12 for infants 5-36 months of age, visual reinforcement audiometry (VRA) is appropriate,13 and children over 30 months can be tested using conditioned play audiometry.14
Daily Use of Hearing Technology
For optimal results, advanced hearing aid (and related) technology needs to be fit as early as possible to minimize auditory deprivation and to assure adequate auditory brain development. Further, advances in parallel processing and “intelligent” automatic circuits have revolutionized hearing aid fittings. Frankly, many of the previously established “rules of thumb” need to be re-examined in light of new insight from luminaries and outcomes-based publications, which indicate multiple benefits from extended bandwidths, maintenance of spatial cues, adaptive noise reduction and directional circuits, and the use of FM systems and remote microphones (see reviews by Dillon15 and Northern16).
Technology needs to provide access to normal conversation and soft conversation, and to the primary speech signal, even in the presence of competing noise. Of significant importance, technology needs to be worn all waking hours. McCreery et al17 reported the results of a multicenter study that indicated 55% of hearing aids were under-fit by at least 5 dB, resulting in insufficient auditory access.
Walker et al18 surveyed parents to determine how many hours their children actually wore their technology. They reported children wore hearing aids an average of 10.48 hours on weekdays and 9.96 hrs on weekends. Of the 272 families, 2 families reported their children never wore hearing aids during the week and 7 reported their children never wore hearing aids on the weekend. Children whose mothers had a college degree had significantly more hearing aid use time than mothers with other educational backgrounds.
Walker et al18 reported parents may overestimate the number of hours of use by several hours per day. Ching et al19 reported the long-term outcome of hearing aid fittings was associated with 1) maternal education level, 2) early functional performance, and 3) the presence of additional disabilities.
Verification and Validation of Hearing Technology
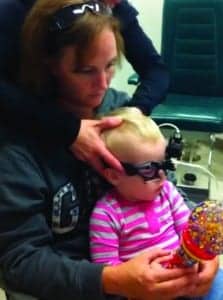
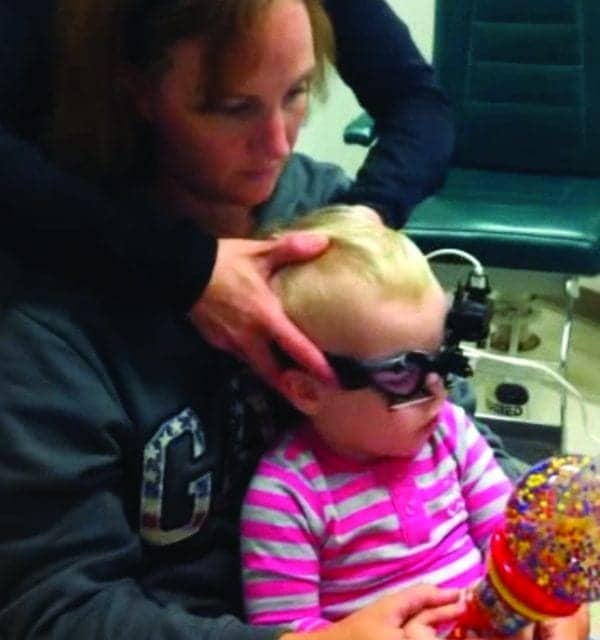
Figure 1. One example of a young child wearing the Interacoustics EyeSeeCam goggle for vHIT testing.
Validation and verification of pediatric hearing aid fittings is mandatory to assure technology is providing appropriate auditory brain access. Validation using real-ear measures (REM) is step one in pediatric hearing aid fittings.
REMs offer exclusive information that is unattainable using any other protocol. REMs are of enormous importance as they are the only physical measure of sound delivered to the cavity between the medial end of the hearing aid and the tympanic membrane. Therefore, REMS are of enormous importance in the validation protocol, but REMs are not the final step. REMs are an important part of the information gathering process, but they do not stand alone as the decisive factor with regard to an appropriate fitting. Importantly, Beck and Nilsson20 noted, “REMs cannot tell you what the patient perceives, indeed one gets the same REM from a dead patient!”

Figure 1b. Another example of a young child wearing the Interacoustics EyeSeeCam goggle for vHIT testing.
For the child using hearing aid technology that is not providing sufficient auditory brain access, there will likely be significant negative effects related to speech, language, literacy, and multiple social skills development. Aided thresholds provide critical information as to whether technology is providing enough access and provide information about how technology settings can be adjusted to provide improved auditory access.21 Speech perception testing at normal and soft conversational levels in quiet, and in competing noise, will provide additional information about how well a child is hearing and will provide direction about audiologic management and selection of educational settings.22
Determining When Cochlear Implantation Should Be Considered
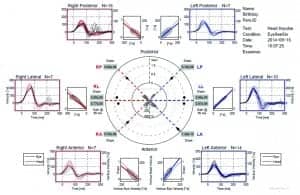
Figure 2. An EyeSeeSix report (Interacoustics EyeSeeCam vHIT) showing the results from a 10-month-old child with normal functioning left SCCs and reduced gain and catch up saccades on the right side.
Validation and verification of hearing aid technology provides information about whether a child is receiving sufficient auditory access. If hearing aids are not providing sufficient hearing for soft speech, if children are dependent on FM systems to hear, if word recognition is not good (defined as 80% or better), or if a child is not attaining 1 year’s progress in speech, language, and listening skills in 1 year’s time, the child with severe or profound sensorineural hearing loss should be referred for a cochlear implant evaluation.
Management of Hearing Loss
Early identification and intervention has an “outcomes-based” positive impact on speech and language outcomes.23, 24 Without appropriately fit hearing aids worn all waking hours, and with lack of access to language and early intervention, children with hearing loss will fall behind their typical hearing peers in language, cognition, literacy, and social-emotional development. While appropriately fit technology is of enormous importance, it is not sufficient for success for children with hearing loss.
For children to develop typical listening and spoken language, they need to be exposed to speech and language at comfortably loud levels (essentially) all day. Parents need to be taught to provide auditory-based language stimulation, and children need vast exposure to language, literacy, and opportunities to develop maximal socialization and communication skills.
Vestibular and Balance Disorders in Children with SNHL
In children, vestibular and balance disorders are highly associated with SNHL; however, most professionals do not routinely screen for vestibular dysfunction among children with SNHL. The prevalence of vestibular and balance disorders in children is frequently underestimated, and it may range up to 15%.25,26 Estimates indicate that perhaps 70% of children presenting with SNHL have impairment of their vestibular system, with 20-40% having severe bilateral vestibular loss.5,27-30 Caregivers (including parents) may not appreciate the relationship between hearing loss and balance, and they may not spontaneously offer their observations or concerns regarding their child’s balance issues unless prompted by an astute clinician.
Vestibular Impairment and SNHL
Not all children who have impairment of their peripheral vestibular system will complain of vertigo. In fact, a great majority of children with vestibular dysfunction will never be vertiginous. This is particularly likely if their vestibular dysfunction is non-progressive, severe, congenital, and/or bilateral, as is frequently the case in association with significant SNHL.
How to Identify a Child at Risk of Vestibular Impairment?
Given a combined cochleovestibular loss in a pediatric patient, impairment of the vestibular end organs translates primarily into impairment of balance. These same children will often have delayed motor milestones. They may demonstrate poor head control beyond 6 weeks, delayed independent sitting beyond 9 months, and/or delayed walking beyond 18 months. On occasion, the child may be labeled as “clumsy” or is perhaps more prone to fall or injury than his/her age related peers. Some children with cochleovestibular loss are unable to perform common childhood activities, such as riding a bicycle without training wheels. The clinician who inquires about motor milestones in a child presenting with SNHL may “open the floodgates” when, indeed, there is a true cause for concern.
Simple maneuvers, such as having the child stand on one foot, can also screen for balance impairment. Specifically, a 2-and-a-half year old should be able to transiently stand on one foot, a 3 year old should be able to maintain this posture for 2 seconds, a 4 year old for 5 seconds, and a 5 year old for 10 seconds. Any child with unexplained delay in motor milestones or an inability to stand on one foot should be evaluated for vestibular impairment (described below) particularly if there is associated hearing loss.
Which Etiologies of Hearing Loss Are Most Likely Associated with Balance Impairment?
Despite modern diagnostic tools and protocols, we remain unable to identify which of the numerous possible etiologies underlies the hearing loss in many affected children. Specifically, many identifiable etiologies of hearing loss have well-described associated vestibular impairments. These include incomplete partition (type 1 to type 3), enlarged vestibular aqueduct syndrome, and more. In these children, the vestibular impairment (similar to the hearing loss) may be progressive in nature. When progressive vestibular impairment occurs, these children may present with true vertigo, which can be severe and may last days or weeks. Vestibular impairment may occur with associated progression of hearing loss or independent of changes in hearing.
Figure 2. An EyeSeeSix report (Interacoustics EyeSeeCam vHIT) showing the results from a 10-month-old child with normal functioning left SCCs and reduced gain and catch up saccades on the right side.
The most important genetic diagnosis associated with vestibular impairment is Usher syndrome (US). Type 1 US presents with congenital onset SNHL and vestibular impairment in addition to progressive blindness due to retinitis pigmentosa (RP). The multi-sensory impairments that affect these children carry with them important considerations for communication strategies over the long-term. A diagnosis of Type 1 US can also be elusive given that it is relatively uncommon, these children show no other outward signs, and the visual loss is slowly progressive and may initially be mistaken for more common visual impairments (eg, near or far-sightedness). In many ways, identification of the associated vestibular impairment in these children may be the key to a more expeditious diagnosis of US. Therefore, we recommend any child with profound SNHL and vestibular impairments should undergo formal vestibular testing and should be referred to a specialized ophthalmologist and geneticist with the specific goal of ruling out US.
A number of congenital and acquired infectious causes of SNHL, such as meningitis and congenital CMV31 infection, also present with associated vestibular impairment. In addition, as our understanding of the presentation, treatment, and hearing outcomes of auditory neuropathy spectrum disorder (ANSD) evolves, it may well be that among the multiple intracranial anomalies associated with more than half of all ANSD cases,32 balance and vestibular problems may be present, yet remain essentially unexplored and undiagnosed.
An additional population of children at high risk for vestibular impairment includes those treated with ototoxic agents, such as aminoglycosides (eg, gentamycin, inhaled tobramycin in cystic fibrosis), chemotherapeutics (eg, cisplatin), and more. Some of these children will be in audiology screening programs due to the risk of hearing loss associated and attributed to these ototoxic agents. Generally, it is less well appreciated that many ototoxic agents are more injurious to the vestibular end organs than the cochlea. Further, vestibular impairment may occur at an entirely different threshold of exposure than hearing loss and, therefore, vestibular ability and impairment should be evaluated.
Cochlear nerve deficiency (CND) is often determined to be the etiology of unilateral SNHL in children born with unilateral deafness. Clemmens et al33 determined that, of 128 consecutive children with unilateral SNHL, 26% demonstrated CND. Further, for children born with unilateral severe-to-profound SNHL, the incidence of CND was 48%.
We suspect many of the children with CND will also demonstrate vestibular impairment on the ipsilateral side.
Tools To Evaluate Pediatric Vestibular Function
With the introduction of video Head Impulse Testing (vHIT), clinicians have a “child friendly,” relatively easy-to-use, and simple tool to evaluate each of the 6 semicircular canals. During testing, the child wears a small lightweight goggle while sitting by themselves or on a parent’s lap (Figure 1). The child’s task is simply to fixate on a target. Targets vary with the child’s age and may include a smiley face sticker on the wall for an older child, or an active toy that lights up and moves for a younger child.
As the child fixates on the target, their head is quickly, gently, and firmly guided along the three planes of motion: 1) lateral, 2) right anterior/left posterior (RALP), and 3) left anterior/right posterior (LARP). The test takes only a few minutes, generating data about the functional gain of each SCC tested, documenting that the correct planes were tested, and demonstrating the presence of any corrective saccades that occurred. That is, vHIT objectively determines if any of the six SCCs are not functioning properly and allows the recording and measuring of any spontaneous or positional nystagmus.
The vHIT assesses the vestibular ocular reflex during typical head movement, and it is sensitive across the normal range of head motion.
Alternative Vestibular Tests and Frequencies
The vestibular system functions over a wide range of frequencies. Therefore, it makes intuitive and clinical sense to test as many of these frequencies as possible (to replicate the real world) before making a diagnosis and rehabilitation plan for a child with vestibular impairment.
For example, the Caloric Test is sensitive to low frequencies (0.004 Hz) and the Rotary Chair Test is sensitive to low to mid frequencies (0.01 – 1.28 to Hz). To shorten the time associated with the Caloric Test (ie, to make it more “child friendly”), we recommend using a monothermal warm screening test (MWST). MWST offers good sensitivity and specificity (83% and 90%) with respect to the bithermal caloric test in children ages 6 to 14 years.
Evaluating the Otolithic Function—Saccule and Utricle
To evaluate the saccule and the inferior branch of the vestibular nerve, the cervical evoked Vestibular Evoked Myogenic Potentials (cVEMPs) have proven useful, even for very young children. With regard to evaluating the utricle and superior branch of the vestibular nerve, ocular Evoked Myogenic Potentials (oVEMPs) have been recorded in children as young as 1 month old.
Summary
Identifying and managing vestibular, balance, and hearing disorders in children can be challenging. However, the identification and management of these disorders in children has significant implications for their safety and long-term outcome.
As we have witnessed vast improvements in the protocols and tools used to screen and assess children with hearing loss abnormalities, similar advances are becoming evident with regard to vestibular and balance disorders in children. Likewise, we expect early diagnosis and management of balance and vestibular disorders in children to yield the greatest benefit for their long-term success.
References
1. Beck DL, Petrak MR, Smith AG. Overview 2013—Migraine, Meniere’s disease and vestibular migraine. Audiology Today. 2002;25(2):54-56.
2. Casselbrant M. Vestibular migraine. In: O’Reilly RC, Morlet T, Cushing SL, eds. Manual of Pediatric Balance Disorders. San Diego: Plural Publishing; 2013.
3. Nandi R, Luxon LM. Development and assessment of the vestibular system. Int J Audiol. 2008;47:566-577.
4. Beck DL, Petrak MR, Bahner CL. Advances in pediatric vestibular diagnosis and rehabilitation. Hearing Review. 2010;17(11)[Oct]:12-16. Available at: https://hearingreview.com/2010/10/advances-in-pediatric-vestibular-diagnosis-and-rehabilitation
5. Cushing CL, Gordon KA, Rutka JA, James AL, Papsin BC. Vestibular end-organ dysfunction in children with sensorinerual hearing loss and cochlear implants—An expanded cohort and etiologic assessment. Otol Neurotol. 2013;34:422-428.
6. Russ SA, White K, Dougherty D, Forsman I. Preface: Newborn hearing screening in the United States: Historical perspective and future directions. Pediatrics. 2010;126[Suppl 1];5-10. Available at: http://pediatrics.aappublications.org/content/126/Supplement_1/S3.full
7. American Speech-Language-Hearing Association (ASHA). Guidelines for audiology services in the schools. ASHA. 1993;35(3)[Suppl 10]:24-32.
8. Ross DS, Visser SN, Holstrum WJ, Qin T, Kenneson A. Highly variable population-based prevalence rates of unilateral hearing loss after the application of common case definitions. Ear Hear. 2010;31(1)[Feb]:126-33. doi: 10.1097/AUD.0b013e3181bb69db
9. Joint Committee on Infant Hearing. Position statement: Principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2007;120(4)[Oct 1]:898-921.
10. Limb SJ, McManus MA, Fox HB, White KR, Forsman I. Ensuring financial access to hearing aids for infants and young children. Pediatrics. 2010;126[Suppl 1]Aug:S43-51. doi: 10.1542/peds.2010-0354I
11. Madell J, Flexer C. Hearing test protocols for children. Pediatric Audiology: Diagnosis, Technology, and Management. New York City: Thieme Medical Publishers; 2012.
12. Madell J. Using behavioral observation audiometry to test hearing in infants birth to 6 months. In: Madell J, Flexer C, eds. Pediatric Audiology: Diagnosis, Technology, and Management. New York City: Thieme Medical Publishers; 2012.
13. Madell J. Using visual reinforcement audiometry to test hearing in infants 5-36 months. In: Madell J, Flexer C, eds. Pediatric Audiology: Diagnosis, Technology, and Management. New York City: Thieme Medical Publishers; 2012.
14. Madell J. Using conditioned play audiometry to test hearing in children over 2½ years. In: Madell J, Flexer C, eds. Pediatric Audiology: Diagnosis, Technology, and Management. New York City: Thieme Medical Publishers; 2012.
15. Dillon H. Interview with American Academy of Audiology; 2013. Available at: http://www.audiology.org/news/super-directional-hearing-aids-noise-reduction-and-apd-interview-harvey-dillon-phd
16. Northern J. American Academy of Audiology interview with Jerry Northern, PhD; 2014. Available at: http://www.audiology.org/news/pediatric-audiology-and-hearing-children-interview-jerry-northern-phd
17. McCreery RW, Bentler RA, Roush PA. Characteristics of hearing aid fittings in infants and young children. Ear Hear. 2013;34(6)[Nov-Dec]:701-710. doi: 10.1097/AUD.0b013e31828f1033
18. Walker EA, Spratford M, Moeller MP, Oleson J, Ou H, Roush P, Jacobs S. Predictors of hearing aid use time in children with mild-to-severe hearing loss. Lang Speech Hear Serv Sch. 2013;44:73–88.
19. Ching TYC, Dillon H, Hou S, Zhang V, Day J, Crowe K, Marnane V, Street L, Burns L, Van Buynder P, Flynn C, Thomson J. A randomized controlled comparison of NAL and DSL prescriptions for young children—Hearing aid characteristics and performance outcomes at three years of age. Int J Audiol. 2013;52:S17-S28.
20. Beck D, Nilsson M. Speech in noise testing: A pragmatic addendum to hearing aid fittings. Hearing Review. 2013;20(5):24-26. Available at: www.hearingreview.com/continuing-education/21662-speech-in-noise-testing-a-pragmatic-addendum-to-hearing-aid-fittings
21. Dillon H, Ching T, Golding M. Hearing Aids for Infants and Children in Pediatric Audiology: Diagnosis, Technology, and Management. New York: Thieme Medical Publishers; 2012.
22. Madell J. Evaluation of speech perception in infants and children. In: Madell J, Flexer C, eds. Pediatric Audiology: Diagnosis, Technology, and Management. New York City: Thieme Medical Publishers; 2012.
23. Carney AE, Moeller MP. Treatment efficacy: Hearing loss in children. J Speech Lang Hear Res. 1998;41(1):S61–S84.
24. Yoshinaga-Itano C, Sedey AL, Coulter DK, Mehl AL. Language of early- and later-identified children with hearing loss. Pediatrics. 1998;102(5):1161–1171.
25. O’Reilly RC, Morlet T, Nicholas BD, Josephson G, Horlbeck D, Lundy L, Mercado A. Prevalence of vestibular and balance disorders in children. Otol Neurotol. 2010;31(9):1441-4.
26. McCaslin DL, Jacobson GP, Gruenwald JM. The predominant forms of vertigo in children and their associated findings on balance function testing. Otolaryngol Clin North Am. 2011;44(2):291-307, vii.
27. Buchman CA, Joy J, Hodges A, Telischi FF, Balkany TJ. Vestibular effects of cochlear implantation. Laryngoscope. 2004;114(10)[Pt 2 Suppl 103]:1-22.
28. Cushing SL, Papsin BC, Gordon KA. Incidence and characteristics of facial nerve stimulation in children with cochlear implants. Laryngoscope. 2006;116(10):1787-91.
29. Cushing SL, Chia R, James AL, Papsin BC, Gordon KA. The Vestibular Olympics: a test of dynamic balance function in children with cochlear implants. Arch Otorhinolaryngol. 2007;134(1):34-38.
30. Cushing SL, Papsin BC, Rutka JA, James AL, Gordon KA. Evidence of vestibular and balance dysfunction in children with profound sensorineural hearing loss using cochlear implants. Laryngoscope. 2008;118(10):1814-23.
31. Zagloski O. Vestibular-evoked myogenic potentials and caloric stimulation in infants with congenital cytomegalovirus infection. J Laryngol Otol. 2008;122:574-579.
32. Beck DL. Reflections on auditory neuropathy spectrum disorder. Op/Ed for the American Academy of Audiology. Available at: http://www.audiology.org/news/reflections-auditory-neuropathy-spectrum-disorder-2013
33. Clemmens CS, Guidi J, Caroff A, Cohn SJ, Brant JA, Laury AM, Bilaniuk LT, Germiller JA. Unilateral cochlear nerve deficiency in children. Otolaryngol Head Neck Surg. 2013;149(2):318-325.
Douglas L. Beck, AuD, is director of professional relations at Oticon Inc, Somerset, NJ; Michelle Petrak, PhD, is director of clinical audiology at Interacoustics A/S, Assens, Denmark; Jane R. Madell, PhD, is director of Pediatric Audiology Consulting, New York City; and Sharon L. Cushing, MD, is director of the Cochlear Implant Program at the Hospital for Sick Children, Dept of Otolaryngology Head and Neck Surgery, at the University of Toronto.
Citation for this article: Beck, D, et al. Update 2015: Pediatric Vestibular, Balance, and Hearing Disorders. Hearing Review. 2015;21(1):14.