BY Douglas L. Beck, AuD, and David P. Speidel, MS
Why AMER is so important and how you can use it.
When sound pressure reaches a certain (and variable) threshold loudness level in a normal healthy auditory system, a sensory-driven afferent signal is sent to the brainstem, which initiates an efferent ipsilateral and contralateral contraction of the stapedius muscle within the middle ear(s). This particular acoustically driven middle ear reflex can be measured easily and is useful diagnostically to reveal important information about the status of the entire auditory system.
Indeed, the AMER “threshold” and the presence or absence of the AMER is of tremendous importance, has myths associated with it, and is often overlooked with regard to the vast wealth of diagnostic information revealed from this simple-to-measure and critically important audiologic measurement. In this article, we’ll use the term acoustic middle ear reflex (AMER) to refer specifically to the stapedial muscle reflex measured by hearing care professionals.
AMER Basics
The detailed anatomy of the AMER is beyond the scope of this article. Nonetheless, for the purpose of review, we offer the following brief anatomic overview of the AMER. When a supra-threshold sound is perceived, the afferent auditory nervous sensory system sends a bio-electric representation of these same sounds from the cochlea to the auditory nerve, to the cochlear nucleus within the brainstem. As the bio-electric signal is received at the brainstem, higher centers are alerted, and simultaneous excitation of descending efferent signals to ipsilateral and contralateral pathways initiates the (normal) stapedial muscle reflex contraction.1
Moller2 offered valuable, clear, and concise notes with regard to the AMER, which will be abstracted here: The “acoustic reflex” is not one thing, it is many. Multiple acoustic reflexes are elicited by sounds. The AMER mentioned above is a “threshold response” that occurs in response to a relatively loud sound. However, there are multiple facial and neck muscle contractions that can be measured through electromyography (EMG) in response to sounds. Further, the “startle reflex” occurs in response to sudden-onset loud sound and impacts multiple skeletal muscles.
Generally speaking, in people with normal hearing and acoustic stimuli, the AMER occurs at approximately 85 dB HL, and the strength of the AMER increases as stimulus intensity increases. In humans, the AMER involves the stapedius muscle (mediated by the seventh cranial nerve) exclusively—although in some animals the AMER may involve the stapedial muscle and the tensor tympani muscle (mediated by the fifth cranial nerve).
The AMER threshold response in the ipsilateral ear is slightly lower than the contralateral ear, and the onset time for the AMER is longer for a threshold response and shorter for a supra-threshold response (as stimulus amplitude increases, latency decreases). In some respects, the sound reaching the cochlea is less variable than the sound reaching the tympanic membrane due to “amplitude compression” via the AMER, more or less in the lower frequencies.
AMER Protective Ability
Moller3 reports that, although the AMER may offer some limited protection against exposure to loud sounds (ie, noise-induced hearing loss, acoustic trauma, etc), the protective action of the AMER is unlikely to have developed for this purpose. That is, sounds that are loud enough to trigger the AMER are rare in nature. With regard to industrialized society, we have only produced noises consistently loud enough to cause noise-induced hearing loss (NIHL) since the industrial age (eg, 1750 to 1860)—a rather recent event in terms of human development, unlikely to create a new central nervous system mediated reflex arc.
The AMER in Pediatric Hearing Care
ANSD. Berlin4 reports auditory neuropathy spectrum disorder (ANSD) is present when:
“…the ABR is desynchronized and the OAEs are (or once were) present and normal and the middle ear muscle reflexes are elevated or absent. The most common cause of ANSD is compromised or damaged inner hair cells. The second most common cause is damaged auditory nerve fibers and the third most common cause is damage to both the inner hair cells and the auditory nerve.”4
Further, as the AMER is so important in revealing the status of the middle ear, inner ear, and ipsilateral and contralateral reflex arc integrity, and for the purpose of indicating or diagnosing children with normal or near-normal hearing, AMER should always be tested. Berlin points out there are many people who have ANSD who have never been tested for or even suspected of having ANSD—including some 15% of the hearing-impaired population and likely a higher percentage of people in the severe-to-profound hearing loss category. He reports that many children who have been diagnosed with auditory processing disorders (APD) likely have ANSD masquerading as APD.
The audiogram is of little consequence in ANSD, reports Berlin. That is, although the index of suspicion may well increase as hearing loss increases, ANSD patients (particularly children) often have normal hearing with respect to their puretone audiograms.
Born without a stapedial muscle: An audiological myth! Berlin4 further reports that, as a postdoctoral student at Johns Hopkins, he evaluated almost 800 temporal bones. Of those, every one had a stapedial muscle. He believes the idea of someone being born without a stapedial muscle is very likely an urban myth. That is, when there is no AMER in response to an appropriately loud sound—particularly in an ear that hears “normal” or “nearly normal”—the first thing to rule out is ANSD rather than to presume the ear is missing the stapedial muscle.
Infants and AMER. Some clinicians were (previously) taught one could not reliably obtain AMERs on infants. In order to establish normative AMERs and test-retest reliability, Kei5 reported on 68 full-term neonates who had passed their newborn automated ABR tests, passed TEOAEs, and passed high frequency (1000 Hz) tympanometry evaluations, and then went on to have AMERs evaluated via 1000 Hz tone. The average age of the subjects was 60 hours. As a group, their mean thresholds are reported in Table 1.
Kei5 reports the frequency effect was statistically significant and there were no significant effects attributable to ear or gender. Test-retest was reported as appropriate, and the author reported AMERs can be included within the test battery for the neonatal population. Further, Kei reported high frequency tympanometry in tandem with OAEs and AMER offers important information with regard to ANSD.

|
| Table 1. Normative AMER data for infants about 60 hours after birth, as reported by Kei.5 |

|
| Table 2. Relative dB SPL corrections when the optional AGC is activated for acoustic reflex measurements on the Interacoustics Titan. |
The AMER Pediatric-Adult Dilemma
Ear canal sizes vary tremendously. The differences between neonate and adult ear canals can be a factor of 10 or more. When fitting hearing aids to neonates, one must consider the real-ear to coupler difference (RECD) to assure the ear canal’s volume has been determined or estimated, so as to correct sound-pressure levels to not over-amplify neonates (or children) based on adult targets. Indeed, when the total volume of the ear canal cavity is halved, the total sound pressure level (SPL) increases by 6 dB; if it is halved again, the total SPL could equal or exceed an increase of 12 dB.6
Clearly, it is important to consider the actual physical volume of the ear canal when obtaining AMERs on adults and children, so as to accurately measure and evaluate the AMER threshold. Another consideration, beyond the volume of the canal cavity, is the depth of insertion of the probe tip, with which the measurements are being made.
The AMER Pediatric-Adult Solution
To measure and correct for the tremendous variation in ear canal size, volume, and insertion depth, Automatic Gain Control (AGC) should be incorporated to maintain an accurate probe tone signal in the ear canal (85 dB SPL with 226 Hz) during measurements. Historically, most clinical impedance instruments have not had an AGC feature implemented for acoustic reflex testing. Unfortunately, this very likely explains the observation that neonates, infants, and children appear to have acoustic reflexes 10 to 15 dB lower than adults.
As impedance instruments are calibrated using a standard 2cc coupler, the majority of ear canal volumes will be smaller due to the insertion depth of the probe and especially with the youngest patients (neonates and infants). Without AGC, the reflex activator stimuli in these smaller ear canals would be higher than the referenced calibration value.
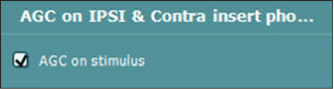
|
| Figure 1. The “AGC on stimulus“ box is located in the Advanced Setup section of the Titan. |
Interacoustics offers a feature called activate “AGC on stimulus” (Figure 1) located in the “Advanced” Setup of the Titan. When this feature is activated, the stimulus level (across all stimulus types) will be reduced by the number of dB as seen in Table 2. For example, when a 0.5 ml ear volume is measured during tympanometry, the stimuli level for acoustic reflexes will be reduced by 12 dB, producing a more accurate reflex threshold measurement.
It is important to note AGC is approximated and, when calculating the number of dB to reduce the stimulus level across frequencies, the same initial calculation (from the AGC adaptation for the impedance measurement) is used across all frequencies. Regardless, utilizing the AGC acoustic reflex function often makes a significant correction that would be otherwise neglected. The correction factor is designed to enable the clinician to use a more scientifically based method for analyzing AMERs.
A Closer Look
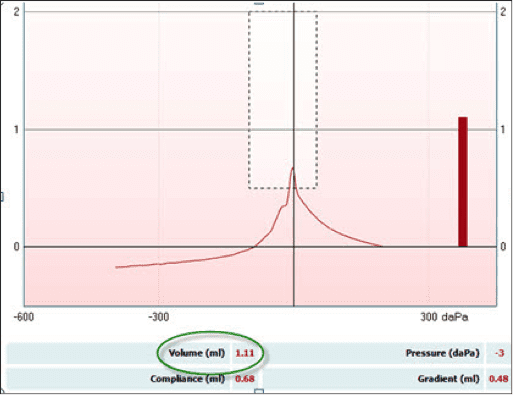
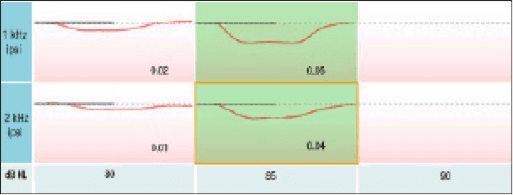
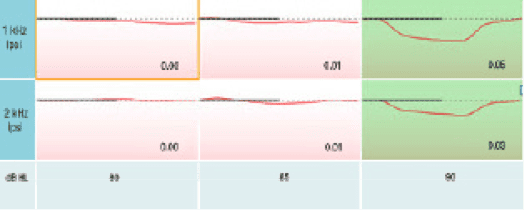
This point is easily demonstrated on a subject with an estimated canal volume (ECV) that is substantially different from the 2cc reference. Note the 1.11 (ml) ECV on this adult subject (Figure 2). This is approximately half the 2cc reference value. When conducting ipsilateral acoustic reflexes on this subject without the AGC enabled (Figure 3), the ipsilateral acoustic reflexes were recorded at 85 dB (dial setting). After turning on the AGC feature (Figure 4), the ipsilateral acoustic reflexes now appear to be elevated and are indeed recorded 5 dB higher at 90 dB.
Apply this scenario to a pediatric subject with an ECV of 0.5 ml and you could expect a threshold shift of 10 dB. In other words, an acoustic reflex previously measured at 75 dB without an AGC function would be corrected to 85 dB with AGC enabled (the true acoustic reflex level!).
Conclusion
The AMER has historically been an important tool for the hearing care professional. Recently, the importance of the AMER has increased dramatically due to its relatively easy and extremely important contribution with regard to the diagnosis of ANSD.
In tandem with a higher profile in screenings and diagnostics, the AMER presents new challenges to the clinician. One challenge is to recognize that the AGC acoustic stimulus adaptation will produce acoustic reflex recordings that are more “true,” but different from what would traditionally be expected from most other instrumentation. Another challenge is reeducating, reorienting, and revitalizing our interest and knowledge of AMER and its tremendous contribution to our profession and the patients we serve.

|
| Douglas L. Beck, AuD, is director of professional relations at Oticon Inc, Somerset, NJ, and David P. Speidel, MS, is director of audiology services at Interacoustics, Eden Prairie, Minn.CORRESPONDENCE can be addressed to Dr Beck at [email protected]. |
References
1. Martin FN, Clark JG. Introduction to Audiology. 8th ed. Boston: Allyn and Bacon Publishing; 2002. Ch 6:153-157; Ch 12:313-345. ISBN 10: 0205366414.
2. Moller A. Hearing—Its Physiology and Pathophysiology. Waltham, Mass: Academic Press; 2000. Section III, Acoustic Reflexes. ISBN 0125042558.
3. Moller A. Acoustic Middle Ear Reflex. In: Hearing: Anatomy, Physiology, and Disorders of the Auditory System. San Diego: Plural Publishing; 2012. Ch 8: 241-255. ISBN 0125042558.
4. Berlin C. American Academy of Audiology Interview with Charles Berlin, PhD [2012]. Available at: http://www.audiology.org/news/Pages/20120809.aspx
5. Kei J. Acoustic stapedial reflexes in healthy neonates—normative data and test-retest reliability. J Am Acad Audiol. 2012;23(1):46-56.
6. Beck DL, Speidel DP, Huijnen J, Petrak MR. Tympanometry and acoustic reflexinnovation, review, and application. Hearing Review. 2009;16(4):28-33.