Strekin AG, Basal, Switzerland, a clinical-stage biopharmaceutical company, announced that the European Medicines Agency (EMA) has granted orphan drug designation for the use of STR001 in the treatment of sudden sensorineural hearing loss (SSHL) or sudden deafness.
Orphan Drug Designation is granted to drug candidates under development that have demonstrated the potential for treating life-threatening or chronically debilitating conditions, which reportedly affect fewer than 5 out of 10,000 people in the European Union, and which lack effective treatments.
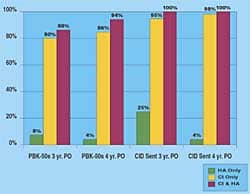
According to Strekin, STR001 is a potent PPAR? (peroxisome proliferator-activated receptor-gamma) agonist currently in Phase 2 clinical development for the treatment of hearing loss. The drug is said to effectively protect auditory hair cells of the inner ear through several mechanisms, and is anticipated to promote hair cell survival and restoration of function in the face of a damaging insult or sudden deafness event. STR001’s otoprotective properties have reportedly been extensively researched in diverse preclinical models of cochlear hair cell protection and hearing loss. Top-line results of the ongoing Phase 2 clinical trial of STR001 are anticipated in the fourth quarter of 2017.
“We are very pleased to receive EMA orphan drug designation for STR001,” said Strekin CEO Alexander Bausch. “This is an important regulatory milestone for the company, and a significant step forward in clinical development of STR001 to fulfill a critical unmet need in hearing loss, for which no approved therapy exists today.”
STR001’s orphan drug designation provides Strekin with various strategic advantages throughout the entire process of development to approval. These include 10-year market exclusivity, exemptions or reductions in regulatory fees, clinical protocol assistance, and access to the centralized authorization procedure for a single application to the EMA, the company said.
Source: Strekin