 Oticon Medical announced that it has obtained 510(k) clearance from the US Food and Drug Administration (FDA) to market the Ponto Abutment Extension for bone anchored hearing.
Oticon Medical announced that it has obtained 510(k) clearance from the US Food and Drug Administration (FDA) to market the Ponto Abutment Extension for bone anchored hearing.
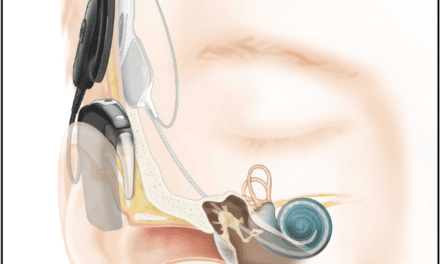
Oticon reports that the new abutment extension seamlessly connects to existing abutments so users can upgrade their bone anchored hearing solutions without changing abutments or undergoing further surgery. The abutment extension provides freedom of choice to all BAHS users. The Ponto Abutment Extension completes the Ponto abutment family, and is a flexible and versatile implant solution.
“We believe that hearing care professionals and users should have the freedom to choose whichever solution best suits their needs regardless of the brand,” said Oticon Medical President Jes Olsen. “The interface compatibility made possible with the new Ponto Abutment Extension means that more people will be able to access the best sound technology available.”
Oticon Medical aims to provide an extensive family of abutments for variations in skin thicknesses. The Ponto abutment concept has features designed to improve some critical aspects of skin penetration for bone anchored hearing. Oticon Medical’s OptiFit and OptiGrip implant design concepts have been cleared by the FDA for use in tissue-preserving surgery. Like all Ponto abutments, the new Ponto Abutment Extension has a medical grade titanium surface.
For more information on the Oticon Medical Ponto System, the new Ponto Abutment Extension, and the family of Ponto abutments, visit the Oticon Medical website.
Source: Oticon Medical





I have a baha abutment and I have had trouble out of two of the same device the baha 5. Now am being told I can’t get a ponto because the ponto will no longer fit the baha abutment any more. I can not have other surgery to just get the ponto so what happened and why was the baha users not informed about this. I feel like I have no other choice now when I did before. Any information would be appreciated.
I bought a Ponto Plus Power. I have a cochlear implant. I need abutment extension. What should I do?