The number of Americans with a hearing loss has “evidentially doubled” during the past 30 years, according to the American Speech-Language-Hearing Association (ASHA).1 Estimates suggest between one and six newborns per 1,000 have congenital hearing loss.1 If caught early (by 3 months, according to KidsHealth.org), some hearing problems can be treated. The Centers for Disease Control and Prevention recommends that all babies be screened before they leave the hospital or before they turn 1 month old. “When infants with hearing loss are diagnosed early and enrolled in early intervention, they have positive speech, language, and listening outcomes regardless of communication modality.”2
Testing the very young, however, is not as easy as testing patients who can both understand and follow direction. The elderly and those suffering neurological disorders may also not be able to indicate when they hear sound and when they don’t. Similarly, these patients may also be unable to participate in the proper fitting of their hearing instruments. Diagnostic systems can provide the voice for these patients as well as increase accuracy in those who can participate.
“I think everyone’s paying close attention to improving efficiency and productivity in the delivery of hearing health care these days, and also providing it in more of a patient-centered way,” says Dave Davis, president and CEO of Otovation, LLC, in King of Prussia, Pa. As a result, audiology diagnostics equipment has gone digital.
PC-based systems have been developed for audiology testing, diagnostics, and fitting. “There are a lot of good reasons as to why this is a good thing for the industry,” says Terry Ross, vice president of MedRx Inc, Largo, Fla. He cites equipment flexibility, size, reliability, and simplicity as both the result of and the stimulus for further development.
“Today, we can measure the actual performance of a hearing device in a specific ear canal and take into account all the variables used in the algorithms—the patient’s age, condition, etc—and accurately and consistently program the device to maximize the hearing aid to the individual’s hearing range,” says Ross. The digital niche, he says, has grown substantially in the United States over the past 18 to 24 months. Hearing Products Report took a look at some of the newest products.
AUDIOSCAN’S HORIZON
“The major driver in our product development tends to be advances in hearing instruments. It was obvious that digital instruments represented a paradigm shift. In response, we developed a new generation of measurement systems incorporating real speech into our Speechmap fitting system,” says Dave Cole, sales and marketing for Audioscan, based in Dorchester, Ontario, Canada.
Speechmap is a hearing instrument fitting environment of the Verifit diagnostic equipment that uses speech signals to map the dynamic range of speech to the residual auditory area. Originally launched with simulated speech signals, the Audioscan Verifit incorporated real speech in 2001.
This year, the company debuted Viewport, a testing suite for fitting, counseling, and verifying the function of digital instruments, including directional function, noise reduction, and feedback reduction. Provided as part of a free software upgrade for all Verifit owners, the four tests verify audibility and the function of digital features.
“Viewport answers the only questions of substance. Is speech audible? Does the directional function work? Does noise reduction behave as it should? Does feedback suppression work? How do all of these features affect audibility?” asks Cole.
The system is easy to use, dispelling the notion that verification takes too long or is too difficult. The tests are integrated and presented on one screen or printout. “It allows the hearing health care professional to fit the aid, verify the function of digital features, and counsel the patient—all in one short session,” says Cole.
Another software feature, one that particularly helps with counseling, is the sensory loss stimulator. Intended for the patient’s family and caregivers, the simulator applies a sensorineural hearing loss to a selection of recorded sounds using the patient audiogram. The program is designed to demonstrate what the patient with the hearing loss is actually experiencing versus simply imitating an earplug.
“For me, a normal hearing person, listening to the sensory loss simulator was a revelation. We’ve had reports from the field of this demonstration moving a patient’s spouse to tears upon realizing what her loved one was really living with. Of course, we aren’t out to make people cry, but this speaks volumes about how powerful a tool this really is,” says Cole.

|
| The Avant A2D+ audiometer’s features include a smaller size; a dual-channel format; integrated Quick-SIN testing; and air, bone, and speech functions. |
MEDRX’S PRESCRIPTION
Patient involvement can enhance a fitting. “We like to engage the patient in the fitting process so they understand what is happening,” says MedRx’s Ross.
The newest designs from the company incorporate some kind of measurement for binaural information that is maximized for the benefit of the patient. The Sure-Probe Microphone system permits monaural or simultaneous binaural measurements.
Audiologists benefit too. Designs are smaller—as small as 5 x 5 inches—and therefore easier to store and move, as well as smarter and easier to use. New lighted probes on the Avant REM Speech+ live speech mapping system indicate which ear is undergoing testing: blue for the left ear and red for the right. The loop design provides easy adjustability to fit any ear size. And an iPodish look provides a little “cool” factor in addition to ergonomic benefit.
The Avant A2D+ audiometer shares a similar modern look and size. The device’s capabilities include a dual-channel format, integrated Quick-SIN testing, and air, bone, and speech functions.
Enhancements to MedRx products are released every 90 to 120 days, “about three or four times a year,” specifies Ross. This year, Ross expects to also see a new tympanometer (“probably in the first quarter of 2009,” he says), a new insert probe (which will also feature the light indicator system), and breakthrough technology in auto-programming hearing devices.
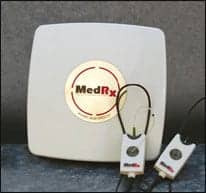
|
| New lighted probes on the Avant REM Speech+ live speech mapping system indicate which ear is undergoing testing: blue for the left ear and red for the right. |
The new technology will allow the computer to actually obtain a frequency target in 12 to 15 seconds. “The computer will calculate the correct performance characteristics of the hearing aid for any target that is entered and will automatically program the hearing aid to that target,” says Ross.
This makes every fitting consistent. “Fittings are within a few decibels of an actual single-frequency target,” says Ross. Users can select the target, either by formula or on their own.
The advance is due to the digital revolution in hearing diagnostic equipment and hearing aids. “Today’s new platforms are far more reliable than traditional, what we call ‘knob-and-dial technology.’ Typically, there are no moving parts, there is plug-and-play simplicity, and the only tether is a USB cable to the computer for power,” says Ross.
OTOVATION’S MOTIVATIONS
“There are a number of interesting trends right now, and two that we’re clearly focused on are mobility and ease of integration with third-party systems,” says Otovation’s Davis. The company’s new OTOPod is a full diagnostic audiometer performing air conduction, bone conduction, and speech testing within a small and portable wireless device.

|
| Otovation’s new OTOPod is a full diagnostic audiometer performing air conduction, bone conduction, and speech testing within a small and portable wireless device. |
Powered by batteries, the device works up to 30 feet away from the PC. “We think that hearing aid dispensers will be increasingly working in the settings where their patients are, rather than the patient coming to them,” says Davis.
Mobile products allow a dispenser to carry a full diagnostic audiometer to places like assisted living facilities, nursing facilities, and patient homes, often tucked into a briefcase/laptop bag. Care is more convenient and personalized for both the patient and the audiologist.
The OTOPod’s speech tests include SRT, WRS (Discrimination), MCL, and UCL, and the device is fully customizable with dozens of testing parameters. An integrated patient response button is built in, increasing patient comfort.
Data can be imported from and exported to other systems, and full-page reports can be easily printed. The 3.5-inch device works with a variety of headphones, including TDH-39, EAR 5A, and Sennheiser circumaurals. “Most people who see the OTOPod are fascinated that a device that’s so small can do so much, and they really appreciate the portability it provides,” says Davis.
The OTOPod, as well as Otovation’s screening product, the Amplitude, both work with Symphony software. The platform is designed to be a hub for all testing and patient record-keeping and can integrate with other systems.
“We’ve done quite a bit of work to enable our products to be easily integrated with other systems, such as balance testing, occupational health, schools, and electronic medical records systems,” says Davis. Full software integration tools are available, as well as a unique test control method that enables third-party software vendors to quickly and easily implement Otovation’s audiometric testing capabilities.

|
| Otovation’s Symphony software is designed to be a hub for all testing and patient record-keeping and can be integrated with other systems. |
The company will continue to enhance the Symphony system, and new features are already in development for 2009. The OTOPod will also get an upgrade, with a new model launching in the spring. And a new two-channel audiometer, the OTOSphere, will debut in the second half of 2009.
VIVA VIVOSONIC
A few months ago (August, to be exact), Vivosonic released the newest version of its auditory evoked response system, Integrity Version 4.5.
“We dedicated the past year to refining the nonsedated ABR [auditory brainstem response] techniques and making the Integrity System more user friendly,” says Yuri Sokolov, PhD, MBA, president and CEO of the Toronto-based company.
New statistical techniques designed for pediatric practices assist in determining the response repeatability and detection within a single test run. Response repeatability is one of the most important indicators of the presence of response, but is often difficult to achieve.
“When a clinician runs the first ABR trace and sees some ‘humps’ on it, she doesn’t know whether those humps are ABR waves and she can stop the run, or [whether] they are residual noise that needs more sweeps to average them out—and this is even more difficult when approaching hearing threshold,” says Sokolov.
The Integrity system reduces the effects of physiological artifacts, and its protection against electromagnetic interferences enables clear results, even within intensive care units and operating rooms. “It’s a nuisance more for the clinicians than the patients, but our system is resistant and not susceptible to interference,” says Sokolov.
For general practices, Integrity 4.5 refined the noninvasive electrocochleography (ECochG) with gold-foiled conductive ear-tip electrodes or tip-trodes. “The ECochG results now achievable with such electrodes are typically as clear as those obtained with needle electrodes and tympanic electrodes, but the procedure is noninvasive, can be administered by an audiologist, and is available to all patients,” says Sokolov.
Since its FDA approval in 2005, Integrity has enabled ABR in infants and young children, as well as other difficult-to-test patients, without sedation and anesthesia. This avoids the associated risks and costs, resulting in better outcomes for the patient as well as the health care organization’s bottom line.
Integrity is marketed around the globe, but was limited by its English-only software. “We are working on internationalizing the software so that it will be easily and seamlessly translatable into other languages, and we are expecting to release this software version in early 2009,” says Sokolov.
Vivosonic expects that current challenges in the economy may delay some equipment acquisitions, but development will continue. “We believe that it is very important that new equipment should not be merely ‘replacement’ of old devices that become unserviceable, but enable clinicians to provide better healthcare services to their local communities on the one hand, and on the other hand, keep up their revenues to withstand the challenges of the economy,” says Sokolov. As the number of hearing-impaired individuals increases, these efforts will be appreciated.
REFERENCES
- American Speech-Language-Hearing Association. The prevalence and incidence of hearing loss in children. Available at: www.asha.org/public/hearing/disorders/children.htm. Accessed October 31, 2006.
- National Center for Hearing Assessment and Management. Utah State University. Diagnostic audiology. Available at: www.infanthearing.org/audiology/index.html. Accessed November 3, 2008.
Renee DiIulio is a contributing writer for Hearing Products Report. For more information, contact [email protected].





