A more reliable method for detecting acoustic tumors
Author’s Note: The goal of this article is to review briefly the rationale, methodology, and the clinical application of the Stacked ABR. For more detailed literature on the Stacked ABR, please see the references at the conclusion of this article.1-5
Click-evoked ABRs have long been used for evaluation of peripheral auditory function and continue to play an important role in evaluating infants and children. An important application of ABRs in adults since the mid-1970s (eg, see Selters & Brackmann6) was for corroborating the diagnosis of an acoustic tumor (vestibular Schwannoma).
Two standard ABR measures—the interaural wave V delay or IT5 delay and the I-V delay—were used. While these measures have shown excellent sensitivity for mid- and large-sized tumors, Eggermont et al7 claimed that small (<1 cm) tumors would often not be detected. This poor sensitivity for small tumors was eventually confirmed in a series of publication during the 1990s, and several of these studies concluded that the use of ABRs to detect acoustic tumors should be abandoned (see review in Don et al2).
Thus, many clinical practices have abandoned the use of ABRs and simply order an MRI with contrast to rule out a tumor. In essence, MRIs are being used as a screening tool because of the failure of standard ABR tests to detect small tumors. However, as my colleagues and I have argued,2-5 there are situations both in the United States and throughout the world where the availability, cost, and comfort of MRI testing are problematic. Thus, it would be of clinical value to have available an ABR test to screen for small tumors.
Revisiting the Usefulness of ABR Screening for Acoustic Tumors
In order for an ABR test to be useful for acoustic tumor screening, it must overcome the failure of the standard ABR measures to detect small tumors. We have hypothesized that the failure of the standard ABR measures to detect small tumors is due to the reliance on latency changes of wave V of the ABR, and that:
1) High frequency fibers dominate the latency measure.
2) Tumors will be missed if these high-frequency fibers are not affected by the tumor.
3) Small tumors do not always affect these high-frequency fibers sufficiently.2,3,5
The Stacked ABR Methodology
A new ABR measure, the Stacked ABR, has been developed and demonstrated, and shows promise for detecting the small tumors that were missed by the standard ABR measures.2 This Stacked ABR measure represents the sum of synchronous neural activity generated from five frequency regions across the cochlea in response to click stimulation and high-pass pink noise masking. The methodology is briefly summarized in Figure 1 (details are found in two other papers2,5), and is based on 8th nerve compound action potential work by Teas, Eldredge, and Davis.9
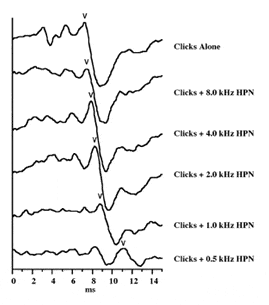
In Figure 1, the top trace represents the response to 60 dB nHL clicks presented alone. The succeeding five traces represent the responses to the clicks presented with ipsilateral masking pink noise high-pass filtered at 8 kHz, 4 kHz, 2 kHz, 1 kHz, and 0.5 kHz. The level of the pink noise before high-pass filtering is just sufficient to mask the response to the clicks. Thus, this series shows the progressive masking of the cochlea to click stimulation. One can observe that, with each lowering of the cutoff frequency of the high-pass masking noise, the latency of wave V of the ABR is progressively delayed. This indicates that the response is progressively dominated from the lower unmasked frequency region and the delay is related to the traveling wave delay down the cochlea.

Figure 2 shows the procedure used to obtain the derived-band ABRs.8 The top trace is again the response to the clicks presented alone. The next five traces result from the successive subtraction of the high-pass responses. These resultant derived-band responses represent synchronous activity initiated from successive octave-wide regions across the cochlea with the theoretical center frequency noted beside each of the derived-bands (eg, 11.3 kHz, 5.7 kHz, 2.8 kHz, 1.4 kHz, and 0.7 kHz). It can also be seen that the latency of the response to the clicks alone is determined by the responses from the highest frequency regions. If each of the derived band waveforms were added together, the sum would be essentially look like the response to the clicks presented alone.
Additionally, by adding the waveforms together, much of the activity from the lower frequency regions of the cochlea will not be seen due to phase cancellation from the higher bands. Therefore, for standard ABR measures, a prolongation of the latency of wave V of the ABR to clicks presented alone will occur only if the tumor affects the high-frequency fibers. In a significant number of small tumors cases, it appears that the high-frequency fibers are not sufficiently compromised such that there is little change in the wave V latency.

To overcome this insensitivity, the Stacked ABR is formed as shown in Figure 3. The Stacked ABR is formed by shifting and aligning the wave V peaks of the derived-band responses shown in Figure 2 (stacking the waveforms), and then adding the waveforms together. In essence, this results in a waveform, the Stacked ABR (last large trace), in which the responses are synchronized across the cochlea.
By aligning the wave V of the derived-band ABRs, the phase cancellation of lower frequency activity is eliminated. Thus, the Stacked ABR amplitude is composed of activity from all frequency regions of the cochlea, not just the high frequencies. Therefore, any reduction of neural activity due to a tumor, even a small tumor, will result in a reduction of the Stacked ABR amplitude.
Figure 4 shows the Stacked ABRs from a patient with a 0.5 cm acoustic tumor. The left panel shows the Stacked ABR from the non-tumor side and the right panel shows the Stacked ABR from the tumor side. The Stacked ABR has been reduced by over 50% by this small tumor.
Effectiveness as a Clinical Tool
Recently, we performed an expanded study on 54 small tumor cases with the Stacked ABR and compared the results to the standard ABR measures.5 The standard ABR measures demonstrated relatively poor sensitivity as shown by many previous studies whereas the Stacked ABR demonstrated that a sensitivity of 95% could be achieved with a specificity of 88% relative to a group 78 non-tumor normal-hearing subjects. Moreover, all (100%) of the small tumors in that study were detected with better than 50% specificity.

FIGURE 4. Stacked ABRs from a patient with a 0.5 cm tumor. The Stacked ABR on the tumor side (right panel) is reduced by over 50% in comparison to the non-tumor side (left panel).
The Stacked ABR measure appears to have sufficient sensitivity and specificity to warrant its use as an initial screening tool. That is, it is sensitive enough to ensure that nearly all patients with tumors are identified and sent for conclusive MRI testing, and specific enough to significantly reduce the number of non-tumor patients sent for imaging. Therefore, the Stacked ABR is a sensitive, widely-available, cost-effective, and comfortable tool for screening small acoustic tumors.
is a research scientist at the House Ear Institute, Los Angeles. |
Correspondence can be addressed to Manuel Don, PhD, House Ear Institute, 2100 W Third St, Los Angeles, CA 90057; email: [email protected].
References
1. Don M, Ponton CW, Eggermont JJ, Masuda A. Auditory brainstem response (ABR) peak amplitude variability reflects individual differences in cochlear response times. J Acoust Soc Am. 1994;96:3476-3491.
2. Don M, Masuda A, Nelson RA, Brackmann DE. Successful detection of small acoustic tumors using the stacked derived band ABR method. Am J Otolaryngol. 1997;18:608-621.
3. Don M, Kwong B. ABR: Differential diagnosis. In: Katz J, ed. Handbook of Clinical Audiology, 5th ed. Media, Pa: Lippincott Williams & Wilkins Publishing; 2002:274-297.
4. Don M. Auditory brainstem response testing in acoustic neuroma diagnosis. Current Opin Otolaryng & Head Neck Surg. 2002;10:376-381.
5. Don M, Kwong B, Tanaka C, Brackmann D, Nelson R. The Stacked ABR: A sensitive and specific screening tool for detecting small acoustic tumors. Audiol & Neurotol. 2005;In press.
6. Selters WA, Brackmann DE. Acoustic tumor detection with brain stem electric response audiometry. Arch Otolaryngol. 1977;103:181-187.
7. Eggermont JJ, Don M, Brackmann DE. Electrocochleography and auditory brainstem responses in patients with pontine angle tumors. Ann Otol Rhinol Laryngol. 1980;75(Suppl):1-19.
8. Don M, Eggermont JJ. Analysis of the click-evoked brainstem potentials in man using high-pass noise masking. J Acoust Soc Am. 1978;63:1084-1092.
9. Teas DC, Eldredge DH, Davis H. Cochlear responses to acoustic transients. An interpretation of whole-nerve action potentials. J Acoust Soc Am. 1962;34:1438-1489.